-
Is this a proper application of sesquation and quotation? My first new non Prime hypothesis. Can it be applied to multivariable equations?
Aluminium forms a sesquioxide Al2O3. Each aluminium has one and a half oxygens combined with it. Similarly cobalt forms a sesquisulphide Co2S3 And so on. There's a joke use of the prefix in the word "sesquipedalianism" The practice of using long and obscure words in speech or writing. Derived from Latin for "a foot and a half long," But when the Romans used the foot as a unit of length, they called it a pes, so I think it's "fake Latin".
-
Units for E = mc² ?
Yep, I know. And both are consistent sets of units. So E=MC^2 works if you are using slugs or poundals.
-
Units for E = mc² ?
I'm just going to mention the slug and the poundal, then run away. Hope people had a nice Easter. https://en.wikipedia.org/wiki/Slug_(unit) https://en.wikipedia.org/wiki/Poundal
-
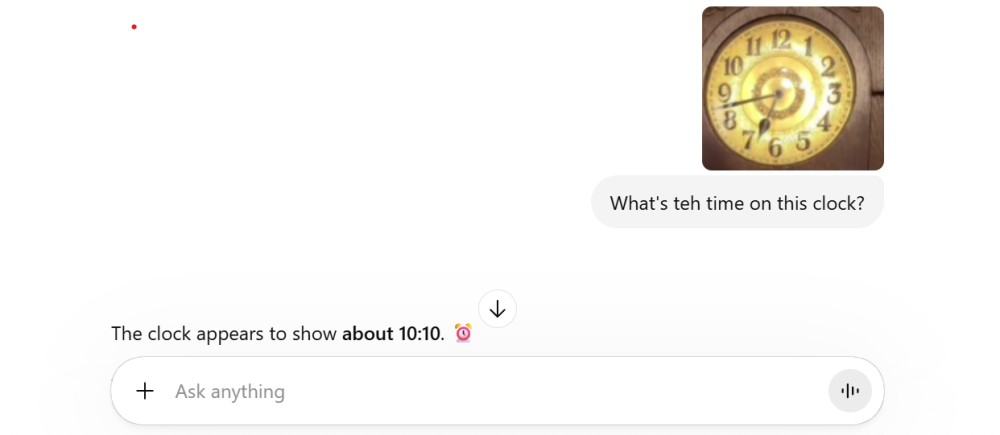
"Wave if you're human"
It gather AI (specifically chat GPT) fails a test that... isn't thought of as difficult. (See pic) But it's the confidence that worries me. As for "scheming" and "deception", all AI's seem to do it all the time. If I tell you that your neighbour has a black cat I might be right or wrong. And I might be wrong, because I'm honestly mistaken. But, given the nature of this interaction, not only do I know nothing about the (hypothetical) cat, I should recognise that I know nothing about it. So, for me to make a statement about it, with the implication that I have some knowledge of the cat, is a lie. I'm effectively dishonestly claiming to know about the cat. And that is still a lie, even if your neighbour actually has a black cat. And AI tells that sort of lie all the time.
-
Today I Learned
TIL You carry just 1 dead body, and that's all you get remembered for.
-
Periodic table group numbering
"It's interesting to group silicon with both germanium and titanium." Chemically, they probably have more in common with each other than with carbon or lead. I wait for Iupac, having decreed in favour of sulfur, to set their sights on "fosforus". While we are at it, "helium should be "helion" or something, because it's not a metal.
-
Underwater lidar...
Can you see the bottom of the sea? If you can, then LIDAR would work. If not, then... not.
-
Could Andrew Mountbatten Windsor be on the Autistic Spectrum?
Indeed, but should we just let him choose bc he is entitled to do so? I didn't suggest that we should. I was pointing out that, while it's possible, it seems very unlikely. The OP's question was "could-andrew-mountbatten-windsor-be-on-the-autistic-spectrum?". Well, he could be. But I doubt it. And if he were , that wouldn't be a "get out of jail free" card.
-
Could Andrew Mountbatten Windsor be on the Autistic Spectrum?
It's possible that Andrew can't tell right from wrong. But, if that's the case, how come he was allowed to fly military helicopters? How did he serve as an ambassador? Seriously, we all know autistic people- and some of us are in that group. In what universe would you employ us for our diplomatic skills? On the other hand, who would be surprised that "Randy Andy's" exploits would get him into trouble? Yes, he counted some pretty evil people as friends. Unfortunately, people typically form friendships with people who they have lots in common with. And he's a rich guy with no responsibilities and nobody to say no to him. That gives him a lot in common with a whole bunch of dictators and "nice" dictators are in short supply.
-
Messages to the president...
Mr President; someone has suggested a name for the new Ballroom. https://en.wikipedia.org/wiki/Orangery
-
Messages to the president...
Please spare a thought for the Translator at Davos who had to translate "If it was not for me, you would all be speaking German" into German. Also, well done to Democratic president Harry S Truman for seeing this potential issue in 1951 and striking a deal with Greenland.
-
Today I Learned
I must have been busy 9 years ago so I missed the opportunity to post this. https://www.youtube.com/watch?v=YaGdwfykYGY
-
Trump Says U.S. Will ‘Run’ Venezuela
Could someone do me a favour please? Tell Trump that North Sea oil has pretty much run out, so there's no point in invading the UK. Thanks. (on behalf of about 70 million of us) And , now for the science part... When North sea oil came on stream, Venezuelan oil actually became more valuable. As you say, Venezuelan crude is heavy. On the other hand, North Sea crude is much lighter. By blending them, the oil companies could use the mixture in their refineries that were designed for use on Arabian oil which is intermediate between the two.
-
Trump Says U.S. Will ‘Run’ Venezuela
It's clear that Venezuela has been gravely mismanaged for decades. And now, they will get "run" by a man who managed to bankrupt six casinos and thinks you can drop drug prices by 800%. I guess the good news is that the people are used to being shafted. It's also fair to say that, if Trump's claims about drugs are honest, the USA will no longer have a drug problem. Is anyone taking bets on that ?
-
Optimal location ?
The sag left to right will be bigger than the sag from top to bottom anyway, because it's a longer unsupported span. If it matters, you should address that first.

John Cuthber
Resident Experts
-
Joined
-
Last visited