Everything posted by studiot
-
Hardness scales...
That may be because hardness is a much less well defined concept, especially for ductile materials and even more especially for very ductile materials such as plastics. Is the required information for machine workability ?
-
For Moontanman and all the Canadians here.
Well I thought perhaps Moontanman and the Canadians was a pop group and I was looking for their new single. 🙂 Really though, I hoped this news would brighten the day for as many persons as possible, and I'm pleased to see it has has some of that effect.
-
Does somebody study complex energy particle ?
My apologies to Mr Stokes for my mispelling of his name.
-
The Physicist and the Philsopher:
Exactly. So why do the Philosophers get it so much more in the neck that the Physicists ?
-
Can someone explain Van Der Waal/London forces to me as simple as possible? Along with explaining Permanent dipole-dipole forces?
What is a positive dipole ?
-
Does somebody study complex energy particle ?
What is all the fuss about ? Mass is not an 'invariant, but sometimes depends upon the environment as well as the frame. Stoles invented 'negative mass' early in the 19th century for the dynamics of soap bubbles rising through water.
-
The Physicist and the Philsopher:
And what do you think of the caloric theory ?
-
For Moontanman and all the Canadians here.
Some local news form near me. Indian relics returned https://www.bbc.co.uk/news/uk-england-devon-61510755 Exeter museum in 'historic' handover of regalia to tribal leader
-
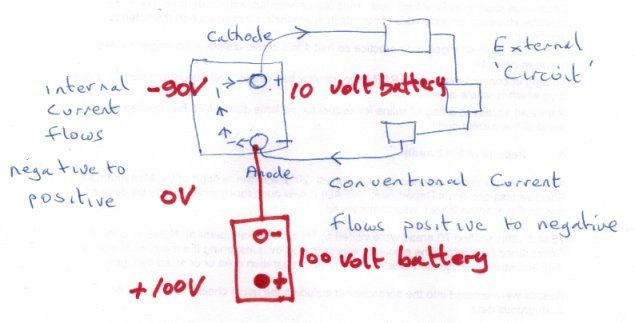
HOW BATTERIES WORK?
You keep referring to the positive (or negative) electrode as though positive and negative were absolute measurements. They are not they are relative statements. Positive relative to what ? Negative relative to what ? I have added a second battery to my sketch connecting its 'negative' terminal to the negative terminal of the first battery. If the first battery has a voltage of 10 volts and the new battery has a voltage of 100 volts and I measure from the positive terminal of the new battery as shown then *) The voltage at the combined 'negative' terminal is -100V and the voltage at the 'positive' terminal of the original 10V battery is - 90V So is the positive terminal positive or negative ?
-
HOW BATTERIES WORK?
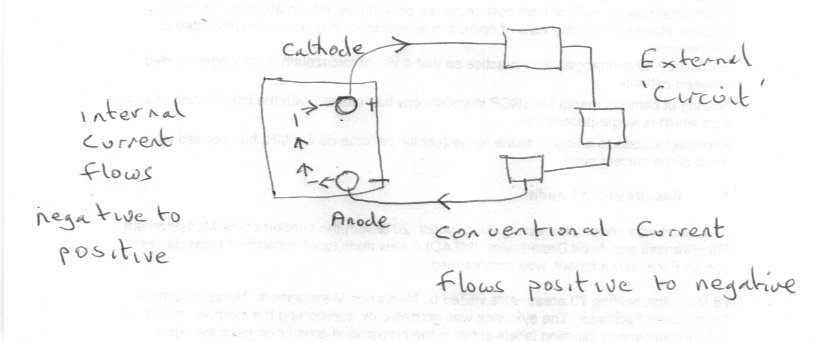
First let me warn you that you have 5 posts total in your first 24 hours. So think before you use the last one and come back tomorrow if you need to. This is not personal, just a very effective antispam measure, and you are very welcome. OK so more electronics than chemistry. Take a device with two terminals, like your battery, with one labelled positive and one labelled negative. Connect up some external circuit. By itself the external circuit does not make a complete loop, although it is called a circuit. The interesting thing is that we choose a direction of current flow (called conventional current) from positive to negative. So the only way for a complete loop or circuit is for the current to flow from negative to positive inside the battery! This is the reason why I defined the anode as the terminal where the electrons leave the device to the external circuit. It is interesting to compare this with the elctronic definition of the anode and cathode for a device such as a diode. Note the electrons flow in the opposite direction from conventional current. OK so your question is basically how does this work out inside the battery. This has been answered here several times actually, in different ways, but the answer is chemistry not positive-negative attraction and repulsion. Where you have a lot of ions in solution they will spread out so that ions of the same polarity will not congregate together but try to get as far (on average) away from each other as possible. Clearly if there are lots of negative ions in solution that will also discourage further negative ions going into solution. But where the negative ions are being discharged their concentration will be below average so negative ions from the rest of the solution will migrate towards the region of discharge due to the concentration gradient. (This is what exchemist was refering to when he said that from the solution point of view the anode is actually less negative). This will in turn reduce the concentration elsewhere, allowing more negative ions to be created at the other terminal, keeping the flow going (which is what chenbeier was referring to) Try to follow the argument through on a diagram like mine and see if that helps.
-
HOW BATTERIES WORK?
The hydroxyl ions are in solution. They are in a paste, but still in solution. So they spread out from where they are generated and their concentration is high, (as do all ions in solution), towards where they are discharged which happens to be the internal part of the anode. It would be helpful if you would tell us your interest in this as we don't know how much chemistry or electric circuit theory you know. There are some misunderstandings/mismatches between the two when you follow round a complete circuit that confuses many.
-
HOW BATTERIES WORK?
Because the chemical reaction at the terminal marked negative on the battery is the anode and that is where electrons are released to travel around the external circuit. In all electrical working the anode is the terminal where the electrons leave the device (in this case the battery) The anode reaction is Zn + 2(OH)- = ZnO + H2O + 2e- So the electrons travel from negative around the external circuit to the positive terminal.
-
The Physicist and the Philsopher:
Hello beecee. as this is your thread I suppose I should ask you what you mean by Physics and Philosophy, and why is this (science) news ?
-
Do somebody study negative energy particle ?
If that is the case are talking about inertial mass or gravitational mass ?
-
Do somebody study negative energy particle ?
Is there any need to shout ? I note that you have degraded my 'isolated single particle' to any old single particle. Single particles undoubtedly do exist, lots of them, as far as anything can be said to exist. That is how they can form systems of single particles. Some have them have zero rest mass. What is the kinetic energy of an isolated single particle ? What is its potential energy ? When there is nothing whatsoever around that particle in any direction all the way to infinity. Both questions are meaningless. By the way I did not say that an isolated particle does not exist, I said its energy is meaningless. I accept that English is not your first language, so try to make sure you actually understand what is said to you beofre you start shouting the odds.
-
Do somebody study negative energy particle ?
I am going to start by giving you +1 for understanding that an anti particle does not have negative energy, it has the same kind of energy as a particle. Unfortunately after that good start, your understanding has failed you. exchemist (+1) has explained that is energy not a particle, but a properties of particles. But here's the thing. The energy of a single isolated particle is meaningless. Energy is a property of a system of particles (and perhaps other things as well). Here's another thing. The terms positive and negative have more than one meaning. They are often used to indicate direction. Chemists have no problem with negative energy, when used in this manner. So if a system evolves (gives off) energy (to another system), that energy is classed as negative. And if energy receives energy from another system, that energy is classed as positive. But there is no such thing as a 'particle of energy', negative or positive.
-
The Physicist and the Philsopher:
+1 for not having a ready made on-demand pat view of either Philosophy or Science, especially one set in stone. By the way, how did this thread move from Physics to Science in general ? I would add to this my observation that neither discipline is set in stone either. Hands up those who think either Philosophy or Physics was the same as it was 50 years ago?; 100 years ago? ; 200 years ago? ; 500years ago ?
-
Why did textbooks get so big?
Another book collector. Welcome. Interesting collection range - movles, computer books and maths books. Please tell us more it will be an interesting discussion. But I suggest you start your own thread (good practice for you).
-
Can someone explain Van Der Waal/London forces to me as simple as possible? Along with explaining Permanent dipole-dipole forces?
Is this for examination purposes ? I ask because UK A level exams are coming up and I was preparing some more detail for you but there is a small but important point I would like to clear up. Van der Waals proposed his froces in 1873, before experimental confirmation of either atoms or molecules. London proposed his quantum idea in 1930 to explain the work on inttermolecular forces that had taken plece in the intervening half century. Since then modern Chemistry has gained a vast amount of new data, both the VDW and London explanations have been refined and redefined several times. So it is important to use he definitions and explanations appropriate to your syllabus if this is exam material. The UK A level currently defines VDW as a general label for intermolecular forces and London forces as a special case.
-
item puzzles
Did you say mathematicians or charlatans ? Your photo looks like a luggage tag or other label to me, which suggested the company logo thought. It is definitely not a MedicAlert tag.
-
item puzzles
Suggest you do a trademark search, perhaps start here. https://www.trademarkia.com/logos-in-two-triangles-geometric-figures-and--260513-2-page-1
-
Market Analysis - Is it science?
I think you missed my meaning about changes and patterns. A few years ago I noticed a daily pattern and built up quite an overall gain using your principle above. But each day the small gain was on different shares and, as you say sometimes I lost, but on balance there was a small steady overall gain. This pattern worked well for several years Then one day we had an unexpected big overall drop when nearly all shares dropped and most of that gain was wiped out.
-
Market Analysis - Is it science?
I agree there are patterns, but there are many more underlying reasons and complications that sensei and swansont have mentioned. Theirs is a very simplified analysis. The timing of sudden, and sometimes large, changes is unpredictable and often catches out 'pattern followers'.
-
Market Analysis - Is it science?
Good points, I like the last line especially. +1 But remember also this is a resurrected thread from 2018
-
Market Analysis - Is it science?
I think that there are plenty of applied Physicists, Engineers and Technicians that would strongly disagree with this statement. But welcome to the discussion and SF.