Everything posted by studiot
-
The Existence Boundary: A Foundational Principle in Cosmology and Quantum Physics ?
Whilst this is quite different from your original claim, where exactly in QM is the requirement for any wave function to exist at all ? what is preventing an entire region or universe being totally empty ? Yes there are formulae such as you have quoted if and only if there is a particle with a wavefuntion and those formulae have been found to be quite accurate. But what pray is the wavefunction for such a region ? what exactly is the wavefunction for a non particle in that region ? And further i have already asked where exactly in QM are the theorems, formulae etc which require virtual particles ? Yes QM allows virtual particles and offers a credible mechanism for their operation. But for this you need to go back to Goldstein, and Higgs. And this business of suddenly introducing local and global and trying to wriggle out of admitting your original claim went too far. That claim is rather like two men in a bar. One has finished his beer the other still has some beer in his glass. So he claims there is beere somwhere (in my glass) and there are no such things as empty glasses.
-
Polynomials and Irreducibility exercises
Thanks Interesting that your field now allows (0, 1). 😀
-
The Existence Boundary: A Foundational Principle in Cosmology and Quantum Physics ?
Thank whoever liked my last post. In point of fact, I think the idea refutes your basic premise as follows. Since probability is always strictly less than 1 it is nowhere = 1. This must be so for the inergral over all space to equal 1. This means that every point has a probability that it is empty or contains nothing at all.
-
Polynomials and Irreducibility exercises
x4 + x2 + 1 = (x2 - x + 1) (x2 + x + 1)
-
The Existence Boundary: A Foundational Principle in Cosmology and Quantum Physics ?
Of course it is in the nature of probability, which is a point function, for the following statement The probability of finding a particle at this point is 0.6 to imply That the probability of not finding the particle at this point is 0.4 This of course must allow for the possibility and probability that there is / is not something else there.
-
The Existence Boundary: A Foundational Principle in Cosmology and Quantum Physics ?
This unsupported calim is as wrong as the previous one that you made about wave functions and I challenged. This is a different, weaker statement than the absolute one you originally made once again please respond with a proper chain of reasoning rather than a bald unsupported claim
-
The Existence Boundary: A Foundational Principle in Cosmology and Quantum Physics ?
Are you sure ? What about an infinite potential barrier or the node points in an s orbital ? Thus far you have stated you one 'principle' , and stated without derivation of any sort, that your principle is compatible with existing theory, which has its own derivation that does not depend upon your E theory in any way. You need to show a path or chain of reasoning demonstrating why this E principle is either true or necessary. So stating that empty space must have virtual particles in it is not enough. Existing theory provides the necessary chain of reasoning starting from the principle of least energy for why it may (but does not have to ) contain virtual particles, rather than being full to the brim with red and green striped unicorns.
-
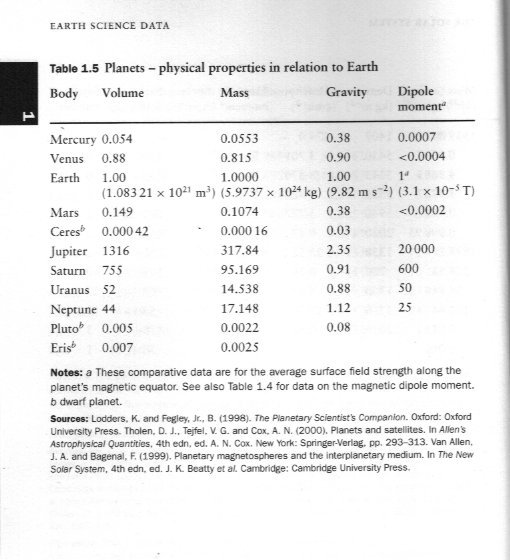
Geo-dynamics core of planet(s) (and presumably stars as well)
The short answer is No. Perhaps you would like to consider how your proposal could be compatible with the following comparative data for various bodies in the solar system.
-
Geo-dynamics core of planet(s) (and presumably stars as well)
Can you rephrase this in English please ?
-
Field Extensions exercises
Interestingly I first thought that the imaginary axis is a counterexample but then I realised that it is not a field, although it does contain Q ! Not sure I accept your proof however since all members of C have the form (a + ib) whilst none of the members of Q have this form.
-
Field Extensions exercises
This makes little or no sense whatsoever. The question can {0, 1} form a field ? makes sense. The question can {0, 1} form a field according to the rules of a different set of numbers ? makes little sense. The rules of forming a field do not include a limitation of the binary operations, but only require the existence of a suitable pair of such operations.
-
The Existence Boundary: A Foundational Principle in Cosmology and Quantum Physics ?
I'm sure we have had something like this before it all sounds very familiar.
-
Field Extensions exercises
2 and therefore mod2 does not exist in this field that has only two members.
-
Field Extensions exercises
this one is a bit tricky and yes can be defined as a (finite) field; it is in fact the smallest field. 0 + 0 = 0 0*0 = 0 1 + 1 = 0 1*1 = 1 1+0 = 0 + 1 = 1 1*0 = 0*1 = 0
-
1. Sub Quantum Echo Particles...(SQEP's) & Sub Quantum Echo Particle Kinetic Resonance Flux
Can I introduce you to the KISS principle ? KISS (Keep it Simple Stupid) https://www.interaction-design.org/literature/topics/keep-it-simple-stupid?srsltid=AfmBOoq5O5di2nqYTSotLvSK7jpCBybGO_1r9STj1NEk99MIoYktDGNE I was reading a history of particle physics and this piece caught my eye. How the simple resonance experiment I have been banging on about with the knotted handkerchief and the hanging weight led Lawrence to the Nobel Physics prize.
-
Unstable Chemistry
If you are looking further into these matters, it would be a good idea to reconsider the phrasing of your question. What you you mean by "... using radioactive decay..." ? Do you mean using radioactive decay of some atom as a cause of the transmutation of itself ? Or are you including radioactive decay of something else to provide radiation as the cause of the decay of some atom that is normally stable ? Thinking about radiation itself is also fruitfull. Radiation comes from to radiate or to spread out in many directions. Clearly when there are only a finite number of paticles radiating, they cannot go in all directions. So studying the pattern of spread is informative.
-
Unstable Chemistry
Note also that strictly the result of both processes is a new atom or atoms and the original atom(s) are used up in the process.
-
Unstable Chemistry
Start here to understand fusion = increase an atom's proton number ; fission = decrease an atom's proton number. Only fission uses radioactive decay for this. https://www.orano.group/en/unpacking-nuclear/nuclear-fission-and-nuclear-fusion-what-you-should-know FUSION FISSION
-
Concrete homes more ecologically friendly than stick built?
I thought that might evoke some comment. 😀 Yes I mean wind loading. We have seen the effect in many recent videos of the storms that are sweeping the planet. Here in Somerset it is interesting to compare my house with that of my immediate neighbour's. My house was built immediately pre WWII, when building largely paused and has concrete roof tiles. My neighbour's was built after WWII when building recommenced and has fired clay roof tiles. The concrete tiles are about twice as thick and heavy as the clay ones and did not suffer in the recent spate of high winds. A significant number of my neighbour's (gosh I wish that word was shorter) caly ones were stripped by the same winds. Another comment about materials. My roof timbers are pitch pine, the resin in which apparantly is very protective from woodworm. Post WWII we started using Canadian softwood timbers in a big way and they are not self protecting and have left owners with a big problem.
-
Making tiny bubbles in very alcaline Portland cement ?
Not sure whether you are looking at the blocks or the mortar for your foamed concrete ? As @LaurieAG says getting the materials science of both of these components right is very important to the performance of the building. Further there are big differences in the requirements for the blocks and the mortar (which does not function as a glue for the blocks, concrary to popular belief). Cowboy builders sometimes add ordinary wahing up liquid to mortar as a cheap easy to obtain alternative to the proper plasticiser. this works OK for the blocklaying time but is deleterious to the mortar in the long term. Yeah Liverpool Catherdral is a famous example of failure to observe proper masonry practice.
-
Evolving Evolution
+1 Perhaps I should mention that I don't give a s___t for Jack Bower's toileting habits. 😀
-
Concrete homes more ecologically friendly than stick built?
My ground floors were originally traditionally suspended timber, but they had rotted and were very draughty So I opted to replace them with a suspended concrete slab. Reinforcement was required for structural reasons. Ready mix offered to 'include the reinforcement' in the form of fibres (carbon ,steel,glass have all been used). I actually went for traditional steel mesh reinforcement. In England the loading codes are more onerous for floors than roofs. Indeed the loading can often be negative or partly negative on a roof.
-
Making tiny bubbles in very alcaline Portland cement ?
Forst of all what do you mean by concrete, ie what is your application ? I ask this because american practice is to use the word cement for what we call concrete on this side of the atlantic. I am replying using the european terminology. Concrete contains inert filler material called aggrgate and binding materials, which are often portland cement as you say. OK so why do you want aerated concrete ? There are three basic reasons for doing this. All use air entrainment (air is the entrained gas, the link gives many types of foaming agents, mostly fancy organic compounds) https://www.sciencedirect.com/topics/engineering/air-entraining-agent Firstly to produce lightweight concrete for structural reasons. Since this concrete is also structural, lightweight aggregate is used along with normal grading. You can even go as far as polystyrene aggregate replacement in suitable parts of the structure. Polystyrene beads are entrained in the mix. Secondly for frost resistance on non reinforced mass concrete. Here larger sized aggregate is often used (40mm and down or even bigger as opposed to 20mm or 10mm and down) as this is cheaper. These would be stones of normal weight. Medium air entrainment would be added at mixing We call this 'bus bay concrete' but I can't see you needing any where you are. Finally air entrainment is used in the concrete to make lightweight thermally insulating building blocks All these blocks are again lightweight and there are various grades. However since the prime purpose of such blocks is insulation rather than structural a variety of pulverised aggrgegates are used, depending upon what is available in the area of manufacture. These pulverised aggregates are often already partly entrained so need less air entrainment with the binder. Does this help?
-
Locked Books
Beware I have a lawyer named Turpin. 😀
-
Evolving Evolution
😀