Everything posted by studiot
-
SFN Migrated and Upgraded
[math]r = \sqrt {\frac{{78.5}}{\pi }} \simeq \sqrt {\frac{{78.5}}{{\sqrt {10} }}} = \sqrt {\frac{{7.85*10}}{{\sqrt {10} }}} = \sqrt {7.85*\sqrt {10} } = 4.98[/math] Yeah, we're getting there +1
-
Ketone?
Your memory is more complete than mine. +1
-
Temporal Substrate Theory: Reframing Gravity and Cosmology Through Time as the Primary Medium”
Here is what is worrying me. And you don't seem to have answered my question, about fields. I can't agree with this. Spacetime (due to Minkowski, not Einstein) is not a field by either definition. In Physicists generally take a Field to mean a region (usually of space) to which a value (that may be positive, negative or zero) may be assigned to every point in that region. For example a temperature field is a scalar field that has the structure you want to describe at every point in say a bar of metal or the atmousphere or whatever. Temperature is the (scalar) field variable to which you can assign divergence, gradients and so on. BUT You cannot 'leave out space' as you have put it. Until this point is clarified I don't see how you hypothesis can proceed.
-
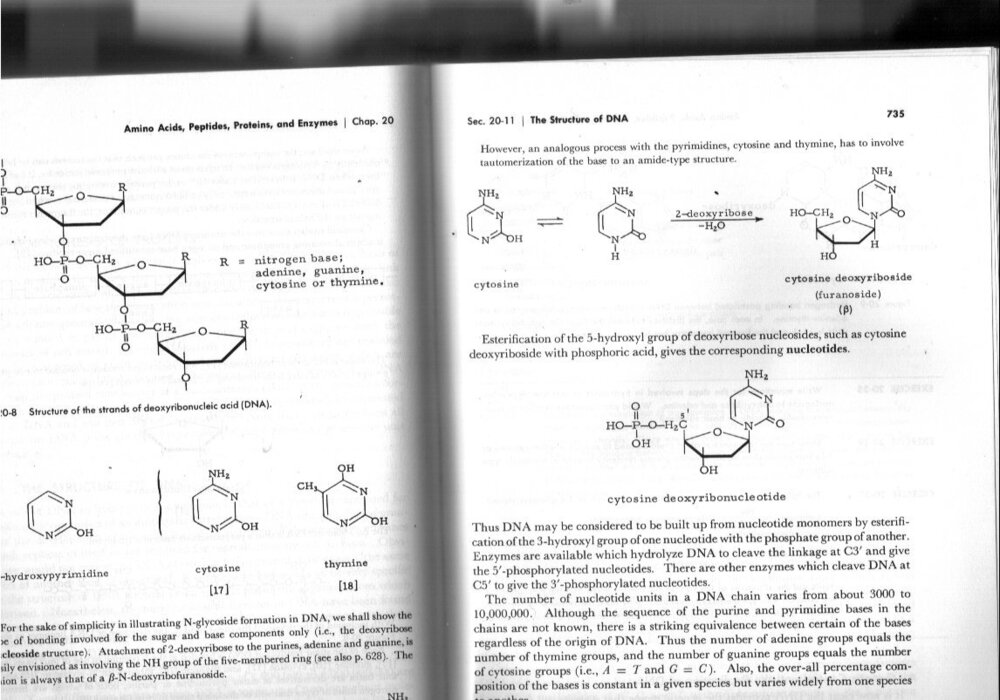
Ketone?
Cytosine has keto and enol forms that rapidly exchange by polytropic tautomerisation. The keto form is the only form shown in the Wiki article.
-
The meaning of constancy of the speed of light
+1
-
The meaning of constancy of the speed of light
No I did not mean Avogadro's constant, since that is directly related to the definition of mass and its associated units. The most fundamental quantity I was referring to is exact. I mean of course, count or number. It takes exactly one oxygen atom and exactly two hydrogen atoms to form one water molecule. Neither a penny more nor a penny less. And this is true whether you are measuring in our solar system, or in the Sirius or Alpha Centauri systems or in a spaceship travelling at some relativistic speed between them. Likewise if you count charges in say an electrolysis reaction. But note counts are not constants, they are just unaffected by electrodynamical considerations. Actually most folks would agree that , despite its flaws and it has some, SI is the most self consistent system of units so far devised. In similar vein you have not understood my comments about the construction of the coordinate system so have answered (when you bothered) inappropriately.
-
The meaning of constancy of the speed of light
I don't misunderstand anything., but thank you for the kindergarten picture. Instead of preaching at me I recommend you ask what I mean if you really don't know. But I suppose as you are not a Chemist you might not.
-
The meaning of constancy of the speed of light
Although this is true of most measurements, it is just not true of all measurements. In particular it is not true of one of the most important SI measurements.
-
Temporal Substrate Theory: Reframing Gravity and Cosmology Through Time as the Primary Medium”
Thank you. So waht is the field variable and what are its units ? Note the EM 'field ' is not one field but two, with two field variables. Out of interest what do you think a field is ? You seem to be using some form of the physics definition, which is quite different from the Mathematics def. I can't agree with this. Spacetime (due to Minkowski, not Einstein) is not a field by either definition. I should be careful of using the word smooth. Smooth has a particular meaning indifferential geometry and function theory, that is very important to this subject, as it is what permits the use of calculus.
-
Temporal Substrate Theory: Reframing Gravity and Cosmology Through Time as the Primary Medium”
Agreed, but note that Tex failure has yet to be fixed.
-
Temporal Substrate Theory: Reframing Gravity and Cosmology Through Time as the Primary Medium”
Hello and welcome as I see you are new here. A couple of words before I address your idea. This forum (SF) has just migrated fro its former host and at the moment many things are topsy-turvey. Some things are working properly some are not and the admins are working hard to correct this. Theoretically a new member is limited to 5 posts in their first 24 hours as an anti spam measure, but I don't know if that is operational. So use your replies wisely. Secondly rule 2.7 requires enough summary for discussion to take place within SF so if you want folks to bite then don't try to send them off site to a link. So I suggest you post a summary of what exactly you mean by a temporal field since you have included space in this definition.
-
Are tangent points allowed in ruler and compass constructions?
OK so I have thought, (sorry my thought processes are so slow at the moment) To do this without a ruler ie with only a pair of compasses you can follow the inscribing a regular hexagon inside a circle, but only mark the hexagon points, not the sides. So 1 ) Centre B draw circle radius BA 2) Leaving compasses set to BA, step off 3 intersection marks round circle from A , and 3 intersection marks going the other way for a check. 3) Find point C on third intersection both ways round.
-
SFN Migrated and Upgraded
One thing you can do to find out who is being quoted is to click on the shadow arrow on the right hand side of the quoted passage. Sorry my screenshot looses my cursor when I take it.
-
New knowledge on a public forum
Yes of course it is possible to reach a conclusion in the way you subsequently defined. That is a particularly useless, possibly misleading situation since that conclusion may still be flawed. I have already given an example of this, which you have not replied to.
-
Dogs V Cats
My brother in law is allergic to cats so that he won't enter a house with cat(s), but fine with dogs and has two of his own. His mother was allergic to dogs, but not cats and had one of her own. So which is better ? Stupid unanswerable question if you ask me.
-
SFN Migrated and Upgraded
Nearly the only useful information on the home page at the bottom right hand corner compared to one of those empty picture holders. The useful things being the who's online list and the clickable all activity (one click instead of two from that diminutive tab at the top)
-
Global Trade wars and the historical significance of such.
Trading rivalry is probably the largest casus belli in history and goes back as far as history itself.
-
SFN Migrated and Upgraded
Nothing has reached me boss.
-
Dogs V Cats
Who is will self ? Have you ever heard of 'ragdoll cats' ? These have more the 'doglike' characteristics you mention. For instance you can take them for a walk. https://en.wikipedia.org/wiki/Ragdoll
-
SFN Migrated and Upgraded
[math]r = \sqrt {\frac{{78.5}}{\pi }} \simeq \sqrt {\frac{{78.5}}{{\sqrt {10} }}} = \sqrt {\frac{{7.85*10}}{{\sqrt {10} }}} = \sqrt {7.85*\sqrt {10} } = 4.98[/math] [math]\left[ {\begin{array}{*{20}{c}} {{E_1}} \\ {{E_2}} \\ {{E_3}} \\ \end{array}} \right][/math]
-
SFN Migrated and Upgraded
Well I don't want to clog up this thread but I couldn't find a way to start my own comment thread. So 1) Notifications, messages and activity list successfully migrated. 2) Login now demands and email address. I really had to scratch my head to find a suitable one. 3) The create table option in the input editor looks good. I will try it out at the next op. Hope it will be so much better than doing it with Tex. 4) Earlier versions used to display what an online user was doing. I found that very helpful in discussion, sometimes, Can it be recreated ? 5) I'm glad to see that the super/sub script options still there, even if it is now two clicks away instead of one. 6) The insert line before quote is really useful, I have often want that on the old one. Haven't yet tried to quote selected text. Can't seem to delete the whole quote though, again a useful thing to do sometimes.
-
Maybe Just My Imagination
Not convinced. Trump is a bully by nature and bullies like threats rather than action as the ation can only happen once, whereas threats can go on forever. Moving military hardware into position is initially a threat, it does not mean it will be used. Unlike Putin, who want to annex a target, China has used such threat to good (for them) effect for a while now in their sphere of influence.
-
Maybe Just My Imagination
Thanks. I'll watch this space with interest.
-
Wall paper remover...
Perhaps favourite responses are because there isn't a one-method-fits-all approach as not all wallpapaers are born equal. So the first task, before stripping, is to determine the type and nature of the 'wallpaper'. In some countries, particularly in Europe (not the UK), it is common to take the wallpaper with you when you move. So obviously the stripping method cannot involve damage to the paper. Indeed special 'strippable' papers are marketed in these countries. Moving on some wallpapers (including the UK) are placed over lining paper or some other lining material such as 'cotina' for either extra insulation or damage concealment or anti-damp or all three. Again obviously you may not want to damage the underlying layer. As already noted, not all glues are the same either. Traditional UK glue is based on plant starch for relative ease of subsequent stripping, but may encourage mould and mildew in damp conditions. More recently modern glues have been available, some designed for ease of stripping, some for the opposite. The paper itself also makes a difference. Modern vinyl wallpaper is impervious to water and often even to steam so iNow's comment about scarrifying the wall is appropriate. Finally, love the pics @Sensei +1.
-
The Official "Introduce Yourself" Thread
Good morning, David. You are very welcome if you have anything useful to discuss or can add anything useful to any existing discussion. It is discussions that are classified in sections, not members; members can access all sections. There are several engineers, and retired engineers here. The official introduce yourself thread is here: Perhaps a moderator would be kind enough to move it there. Please note that in your first 24 hours you are limited to 5 posts as an antispam measure.