Everything posted by studiot
-
First post, hello, I have a lot of questions.
No you are not thick and these are basic questions, which as you say, are fundamental. But they are very good questions. Don't worry we are covering these. I hope you are taking in the new terminology. I am trying not to introduce too many new words at once. So I think we are ready for your first chemical equation ACID plus BASE = (makes) SALT plus WATER Note that chemical equations are somewhat different than mathematical ones, although we will come to do some mathematics with them. Such a chemical equation represents a chemical reaction (or in this case a whole class of chemical reactions) So we have Hydrochloric Acid plus Sodium Hydroxide makes Sodium Chloride plus Water. Acids were once called spirits and hydrochloric acid was called spirits of salt. Sodium hydroxide has a common name as caustic soda. So our friend either made the chlorides this way or bought them ready made. I think MigL has described acids quite well for you. if that is not enough please ask for more. The 'salt' created by hydrochloric acid is called a chloride. Why chlorides ? Well most chlorides are (very) soluble in water. That's how the presenter got the solutions. There are always exceptions the main ones being the chlorides of silver, lead and mercury are hardly soluble. So next bit of theory. Solutions are made by dissolving a solute (sugar, salt etc) in a (suitable) solvent. Thinking back to our earlier posts a solution is a mixture because the parts can be separated by mechanical means boiling off the solvent to leave the solute behind. This process is called distillation. Another similar sort of mixture is called a suspension. Here fine particles of one substances is intimately mixed with another substance. The result is called a suspension. This is not solution, although the suspended particles may be recovered by boiling, they may also be recovered by passing the suspension through a sufficiently fine filter. Finally (for this post) we can also separate the solute (or part of it) by a Precipitation Reaction. Here we take a solution of an obviously soluble salt and add a solution of a different salt which will react with the salt already in solution in such a way as to produce a Precipitate. This happens when the added new solution contains ions that are insoluble as a salt of the original solute. As an example we now know that silver chloride is insoluble, and I am now stating (from experience) that silver nitrate is soluble. So if we add a soluble chloride (such as sodium chloride) to a silver nitrate solution we we observe a (white) precipitate - of silver chloride. In effect the sodium and silver exchange their type of salt. This sort of reaction is important in both analytical chemistry and for the preparation of chemical compounds. The silver chloride can be filtered off or used in a second reaction as in the video. We are now building up chemical vocabulary to use in the future.
-
First post, hello, I have a lot of questions.
The flame test I referred to forms the beginnings of most modern spectroscopy, one of the main modern analytical tools. Chemists have much to thank Physicists for in the development of the modern spectrometer. There are various sorts, Infra red, Mass, Ultra violet, etc. These are capable of answering the "How much ?" question and are often fully computerised and automated. So there is a new term here, spectrum, which basically means a range of values. Note also in the flame tests that the presented is using ions, which we will come to. These are ions in solution (dissolved in a solvent) probably the most common way Chemists use ions.
-
First post, hello, I have a lot of questions.
Ok so let us look further into analytical chemistry. Say we have plenty of sample to play with. We are normally interested in the answers to one or two questions or both. The first question we ask is What is in it or what is it made of ? This is called qualitative analysis and is by far the easier of the two. The second question is How much ? This is called quantitative analysis and I will come to that later. So we can look at it and ask Is it a solid, liquid or gas (at room temperature) ? What colour is it ? - Most substances are a white powder, colours are less usual and generally distinctive. Will it dissolve in water, acid, benzene, or other common solvents ? Geologists carry little bottles of acid which test rocks for the presence of calcium (eg limestone, chalk etc). Lavousier may well have tried a flame test, although he would not have the modern advantages in this video from MIT. Note in the video after the introduction the flame colours are shown with the overall flame colour on the left and the different colours making up the overall on the right.
-
Speculative science questions
Is my screen deficient? Only I can't trace a response to my previous post. Here is some fluid mechanics theory which describes your idea. But you cannot go back before the initial conditions.
-
First post, hello, I have a lot of questions.
Glad you liked it. So I would not start with atoms, molecules, electrons and protons, although you mqy have heard of some of them. This is because we start with our encounter with the surrounding material world. We create words and language to name and describe these things, rather as I have done with iron, wood, concrete. water etc. Then with our scientific hat on we abstract properities, characteristics and abstract descriptions about these same things and how they might be similar or different. We create an abstract model in our heads, computers or on paper. So then we have two worlds a direct material one and an abstract one to deal with and this abstract one is where beginners especially find difficulty. So I am going to suggest you re-read the list of sub caegories of chemistry I mentioned in my first post and we find a few examples in a preferable category to introduce the abstact science of chemistry and its specialist nomenclature. You have already done this to some extent with your question This is the way to keep the theory in touch with the tangible which makes for a more interesting subject experience. So how about you pick one area and I will offer some examples ?
-
This is my documents. Please point out any mistakes and let me know. Please do this to help me, thank you.
Indeed so. +1 Because Your document is not Science. In fact it contains many obvious experimental inaccuracies. Proving anything is not the scientific way. A counterexample may or may not disprove something. Over two and thousand years ago Greek philosophers believed that they could sit in the comfortable villas and dream up hypotheses about how the natural world behaves without ever checking to see if their ideas replicated what actually occurred. They were serious wrong in many many respects and it has taken Science thoudands of years to move on to a better ( as in more successful) approach. Unforunately you appear to be trying to tread the same path as those ancient Greeks. So I seriously you do a couple of things. Firstly find out about the Scientific Method, which drives scientific activity. And also findout the difference between an Hypothesis and a Theory. You can ask here at SF about this, I'm sure Phi is bursting to explain this. Secondly ask about some basic understanding of Science in general and Physics in particular to improve your future thinking.
-
Best Materials for Lightweight Robotic Arm Design?
Good reply +1. Robots are the subject of electromechanical engineering. Is your laboratory some other discipline and you need a robot arm for say moving test tubes ? Or is this an academic electromechanical project ? I ask because if this enquiry is part of a coursework or project work we are only allowed to advise. My thoughts here are that the arm should be hollow, with a sectional material as far as practicable from the holow centre, and preferably longitudinally fluted or ribbed. This will ensure the necessary rigidity as far as possible. The mobile platform should be counterweighted to avoid and tipping when the arm is extended extended past the edge of the platform. A carbon fibre fishing rod will hold 2kg but bend excessively and not be controllable for precise placement. I would suggest that ease of manufacture should be considered, depending upon what you have available. Titanium requires special machines methods. Aluminium is subject to fatigue, depending upon the alloyand is extremely difficult to weld reliably so consider riveted joints. Carbon (or glass) fibre should be laid in mats with the weave laid criss cross to form the hollow tube. The whole process requires curing time etc for the resins so a single arm would take longer to manufacture that metal. This is an important factor in a production line.
-
First post, hello, I have a lot of questions.
OK to start see my reply to this thread Any questions on this ?
-
Fog harvesting could provide water for arid cities
When did you last go up Ben Nevis ? 😀 Dune is Science Fiction which also involved telephathic powers and other stuff. But thanks for the replies guys.
-
Fog harvesting could provide water for arid cities
- Messages to the president...
Just like back home in good old Blighty.- First post, hello, I have a lot of questions.
Most traditional textbooks split upon the lines I have outlined. They often go through a long list of chemical substances and their properties. Although very systematic and thorough this can be rather boring for many. However If you were in the aviation industry you can probably cope with high school maths so this book might be an appropriate overview Feel free to ask about anything you read that needs amplification or explanation.- Messages to the president...
Would these be the same folks who can't 'see' climate change ? 😀 Yay +1- First post, hello, I have a lot of questions.
Yes welcome, here is some starter information that may be useful. Originally Chemistry was divided into two categories Organic Chemistry and Inorganic Chemistry. The word organic meant that the 'chemicals' ( correct word substances ) derived from living things. Everything else was not organic or inorganic. We now know that this distinction is not accurate but these two categories still remain although organic chemistry has been revised to the chemistry of carbon and its compounds. So basically Chemistry is about all the varous types of substances and the interactions between both themselves and non material things such as light and magnetic fields. Since those early days important sub classifications have been made such as Analytical Chemistry the Science of determining exactly which substances there are in a smaple and how much of each. Physical Chemistry The Science of the physical properties of substances such as density, colour, melting point, boiling point, refractive index, solubility etc Structural Chemistry The study of the internal structure of each and every substance Reachion Chemistry the study of rates and mechanisms if chemical reactions. All of these apply to both Organic and Inorganic Chemistry. Yet more subcategories apply to more specific areas such as biochemistry, mineralogy, crystal chemistry, rheology, molecular chemistry etc.- Mass and curvature (split from Question about matter and space-time)
Firstly you seem to me to be describing curvature here and in previous posts in this thread using too few dimensions. When you get to space or spacetime there is no such thing as 'opposite curvaure' . Secondly you might like to review a simpler system, that of the difference between moment of inertia and product of inertia and the inertia tensor. This offers a simpler explanation as to why the are many solutions a given moment of inertia, but the product is unique.- Best Materials for Lightweight Robotic Arm Design?
Hi Jane and welcome. This is presumably a project ? I suggest you haven't given us enough information. You should sit down with your tutor and draft a detailed specification of what you want to this arm to do. Lift - how much ? Transport how far ? environmental conditons of working - temp range -humidity - hazardous atmousphere ? and so on. Having a target objective is the way to a splendid project.- Window air conditioner, reversed.
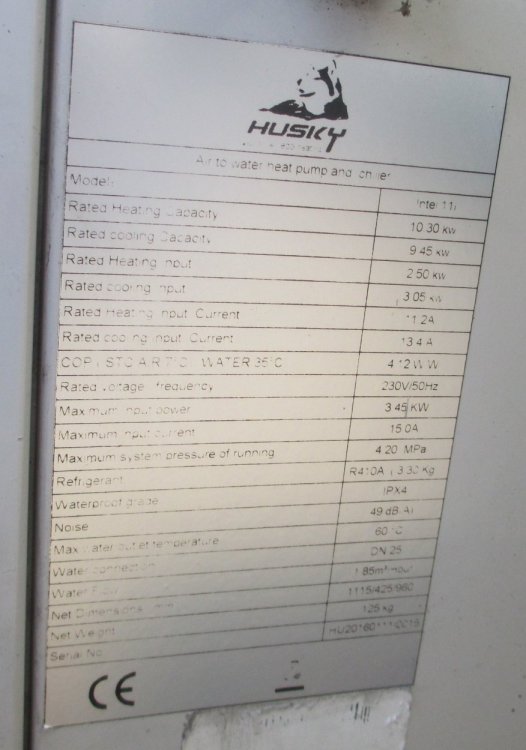
Here is the rating plate on my heat pump. Note it is not the window or protable variety. The cooling mode has about 10% less capacity than the heating mode.- Gap between life and non-life (split from What if god...)
One point I keep making that only swansont seems to appreciate concerns 'precursors'. Most replies seem to assume there is only one 'precursor', which is of course, impossible for building more complex molecules. But not only do we need at least two precursors and reactions, all the evidence suggests that we need a whole chain of them. The important point is that we do not know the length or composition of that chain. These fact pose immense difficulties in estimating overall probabilities and Ken's efforts are far too simplistic. For instance the reaction that produces precursor A may occur in one galaxy and the reaction that produces precursor B may occur in another or at a time beyond the lifetime of the A molecule. And the new AB molecule must have a lifetime long enough to participate in the next reaction for the next stage. The treatment of the volume over which the concentration is calculated is important. So selecting nice concentrations and reactions rates is unviable. If I were asked to suggest a calculation scheme I would suggest assigning partial probabilities to each stage of a probability tree and considering the consequences of 1, 2, 3, 4 ....n stages. This approach is taken in engineering Limit State Analysis and Design and that is complicated enough; the reaction pathway to life almost certainly has many more stages.- Optimizing Solenoid Design
If you are going to put it or them into a card then you need to increase the turns not by the usual linear lineup of one alongside the other but in a flat spiral fashion. In order to mechanically couple this to the effort use a mechanical membrane, rather than a central push rod.- Mass and curvature (split from Question about matter and space-time)
Glad you are back and had the time to compose that excellent explanatory post. +1- Gap between life and non-life (split from What if god...)
Ken I am not getting at you, indeed some of my comments were directed at this statement from CharonY I call your and everyone elses' attention to the following paper, desccribed by Prof Benton as "One of the msot daring papers ever published .... yet one of the msot influential papers of the 20th century" Extra Terrestrial Cause for the Cretaceous - Tertiary Extinction. Nature 6th June 1980 Alvarez , Alvarez, Asaro and Michel. It introduced the famous iridium spike and a formula for the geological effects of meteorite impact. My point is that we just do not have enough information to calculate probabilities so readily. Which is why I offered something we can calculate and swansont kindly amplified. As regards meteor impact there exist very serious barriers to the idea that these relatively delicate 'precursors' arrived by meteorite. Do you have a recipe book by the way ? Meteorites are known to have enough energy to melt and even boil rocks, by the impact, so what would impact do to precursors ?- Gap between life and non-life (split from What if god...)
Ken you also need to consider temperature. At the temp of space (say 3o) or at the more comfortable temperature of an Earthlike planet (say 300o) or at the temperature of a hydrothermal vent (400o to 450o). The Arrhenius equation is very relevant here.- Silly question?
The point about equations is that it does not matter which way round you write them. If A = B then B = A Most of the laws of Physics are of this form so if Time is involved it does not matter whether time goes forward or backwards, the equations still hold. There is one major law of Physics called the second law of thermodynamics which doe not have the form of an equality (or equation), it is an inequality so If A < B then going the other way B is greater than A. Since the second law involves time it cannot be reversed and hold the same form like an equation.- Gap between life and non-life (split from What if god...)
You were very clear, but just plain wrong. It is very far from clear to me. You are just not understanding what you are being told, perhaps that is why you have not responded to my previous post. Let us take a much simpler example of chemical kinetics that we can calculate. The chemical reaction between two of the commonest molecules in the universe. Consider a cluster of hydrogen gas. We can calculate the minimum size required for this to ignite as a fusion reaction, forming a star, or the for the time we would have to wait to expect such an ignition to happen. But we do not have the necessary information to do this the the many times more complicated series of reaction that are necessary for life to occur.- Gap between life and non-life (split from What if god...)
I understand from this claim that you did not actually understand my explanation at all or you would not have said Because in a laboratory you would have control over the concentrations. What I also meant was we do not know what concentrations to control. That follows directly from my description of chemical kinetics. - Messages to the president...
Important Information
We have placed cookies on your device to help make this website better. You can adjust your cookie settings, otherwise we'll assume you're okay to continue.