Everything posted by exchemist
-
ChatGPT
Sure, it might sometimes, but others on this forum have found instances where it is wrong or nonsensical. As @studiot says, it is language model. As such it is not well adapted for mathematical science. You asked if it is reliable, and the answer is evidently no it isn't.
-
The concept of Time
No, north of the North Pole is not space. There is no north of the North Pole. The points of the compass relate to the surface of the Earth.
-
The concept of Time
My understanding of the most popular model is that time itself only started at the big bang, i.e. there was no “before”.
-
Are West Europe countries are second class countries in Today's world?
Oh I don't know. I thought Martinique and la Réunion were pretty good when I visited. And my sister-in-law seems happy in New Caledonia, where she has lived for 30 years or more. But I have not lived in these places myself, admittedly.
-
ChatGPT
You're a mug then, because it isn't. We've already had some threads discussing how unreliable it is for mathematical science.
-
Airlines And Other Private Companies Going Into The Space Industry
What about Space X? Or Jeff Bezos's Bellend One? There is no bar to private companies getting into the business. The reason there are not more is the high cost and risk, which are easier for government to sustain than private companies, backed by commercial banks. And as it is NASA, ESA and the Russian and Chinese programmes compete to offer launches for satellites on a commercial basis, so not all their funding is from government by any means. But it's true that it is a freight rather than passenger business. There are obvious reasons for that. It's very expensive, the life support systems for people would add further cost. And what's the point? There's nowhere to go once you're up there.
-
HOWs this actually happening???
To comment usefully on this we are going to need more than just pencil sketches, with no accompanying explanation. What is it that you have actually seen, and in what context? (I do hope it is not a YouTube video.)
-
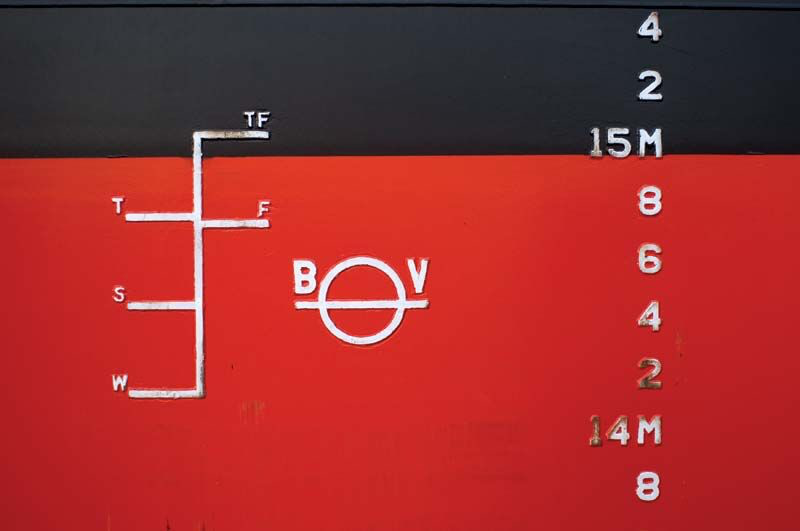
Why icebergs do not sink?
And even that varies from ocean to ocean, hence all the markings on the Plimsoll Line on a ship's hull, showing the different limits to which it can be safely loaded:
-
Why icebergs do not sink?
The density of ice is lower than that of liquid water at the same temperature. Unlike most materials, ice expands upon freezing. Objects that are lower in density than the liquid in which they are immersed will tend to float in it. The reason it expands on freezing is because the molecules in the solid take up orientations that maximise the strength of hydrogen bonds between the molecules. This leads to a more "open" structure than in the liquid. The physics that governs whether and how an object floats is to do with the relative magnitude of the object's weight and the buoyancy force it experiences from the liquid. This is set out in what is known as Archimedes' Principle. There are details here: https://en.wikipedia.org/wiki/Archimedes'_principle
-
Fuel consumption as an area
That may be a good strategy, as one of the issues not much discussed as yet is the need to rewire entire neighbourhoods to cope with the higher electricity demand, once most people have EVs. If people can generate at least a proportion themselves it could help a lot.
-
Fuel consumption as an area
From what I have read, it is better to keep an old IC vehicle as long as you can, before trading it in for an EV, due to the large and unavoidable carbon footprint of manufacturing a new vehicle, of any type. But your next purchase, whenever it is necessary, should be an EV. The longer you can leave it , the greener the EV will be , since the renewable proportion of the electricity it consumes will grow as the years pass. Also the charging network will get better over time. I'm currently running a 19yr old petrol VW Golf, (the 1.4l one with both supercharger and turbocharger, which performs like a 2l but with much better fuel economy). It still works fine and does what I need it to do, so I intend to hang onto it for another year at least.
-
Why not use ZnSO4 for sulphuric acid.
Don't be an arsehole. The procedure you were suggesting could easily put someone in hospital.
-
Are West Europe countries are second class countries in Today's world?
On the contrary, the countries of Western Europe are by far the best to live in, anywhere on the planet.
-
Fuel consumption as an area
Yes. If the rolling road measured the power (torque x revs) output of the engine at the driving wheels, then if you measure the rate of fuel consumption and you know the calorific value of the fuel, you can work out what proportion of the calories burnt end up as mechanical power. It will be 25-30%, I expect. There will be some errors due to frictional losses in the transmission, so you will slightly underestimate the output of the engine itself. In a mechanical transmission those losses are small - <2% if I recall correctly - , but with a torque convertor they may be more significant. There may also be some errors due to the fuel not being 100% burnt to CO2.
-
Why not use ZnSO4 for sulphuric acid.
Indeed. So you should not suggest hazardous procedures without at least pointing out the hazards.
-
Why not use ZnSO4 for sulphuric acid.
Then you should not be recommending this procedure to a person whose expertise you do not know, on a public forum.
-
Why not use ZnSO4 for sulphuric acid.
This strikes me as a terrible idea. Capturing large quantities of these gases is risky enough, but for an amateur to attempt to react them is definitely a recipe for an explosion.
-
Why not use ZnSO4 for sulphuric acid.
How does this work? When you electrolyse NaCl, you evolve H2 and Cl2, and you are left with NaOH, i.e. an alkaline solution.
-
Fuel consumption as an area
Instantaneous consumption is given by the rate of consumption, isn't it? The units of which are volume per unit distance, sure, but it does not seem to me to add anything to express this as an area, even though dimensionally that is what it is.
-
Fuel consumption as an area
Yes I think so. What insight does this division provide?
-
Why not use ZnSO4 for sulphuric acid.
Well the pH shows a value of about 1, with indicator paper, which is very approximate. You would get 1.3 with 0.1M oxalic acid: https://www.aqion.de/site/ph-of-organic-acids. So that doesn't prove much on its own. But it is true that the precipitate looks blue-white, which looks right for copper oxalate. The reaction is referred to here:https://en.wikipedia.org/wiki/Copper_oxalate as a method for making copper oxalate, with sulphuric acid as a byproduct. (This video is by somebody different - a Dutchman by the sound of him. I can't place the earlier one.) If it works with copper sulphate it might work with zinc sulphate, seeing as that too is insoluble in water.
-
Why not use ZnSO4 for sulphuric acid.
Hmm, a YouTuber who makes a point of trying to get round EU laws. Not sure I approve. But purely from the chemistry point of view, if the oxalate is insoluble and precipitates, then that should drive the equilibrium to the right, for a while. As the acidity increases though, less and less of the oxalic acid will dissociate into oxalate ions, so the thing will slow down and stop at some point.
-
Why does fine-tunning for life suggest a multiverse?
I tend to agree. Everything has to have some value and the fact that a constant it has the value it does is no more improbable than any alternative value. To give a trivial example, the fact of my personal existence relies on a highly "improbable" chain of coincidences, regarding how my parents were conceived, how they happened to meet and so forth. But nobody feels the need to argue my existence must have been "designed" by some great plan, because of its intrinsically low probability.
-
Why not use ZnSO4 for sulphuric acid.
OK, understood. Though this seems to be a very roundabout way to make it. Can you supply a link to show us where you are getting this route from? Zn oxalate seems to be insoluble in water so I imagine it might work in the same way.
-
Why not use ZnSO4 for sulphuric acid.
Ah, possibly not. Regarding whether it works, I was wondering about that. If an insoluble chelated metal oxalate is precipitated from an aqueous solution, I guess you are left with an acidic solution with sulphate ions, i.e. dilute sulphuric acid. But it can't be a way to generate pure sulphuric acid.