-
Posts
682 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Everything posted by Horza2002
-
Lol, could you possible write any smaller They seem ok. So where did you hydrogen come from that you needed for the hydrobromination then? And which one did you decide was the lowest energy conformation?
-
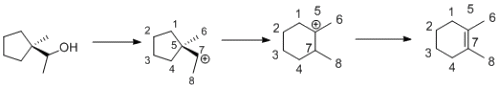
Have a look at the picture I've attached. If you generate the seconday carb-cation as you have demonstrated, then we can do a rearrangement to so that we no have a tertiary carbo-cation. Tertiary carbo-cations are more stable then secondary ones due to the increase in hyperconjugation. I have also number the carbon atoms so you can see where they move too. One of the carbons of the ethyl group has become part of the ring.
-
Firstly, why don't you draw out the structures that you think you would get if it was a bromination. A hydrobromination is a specific type of halogenation (since bromine is a halogen). What do you get if you treat an alkene with bromine?
-
Its not 100% clear, but I think you've now got them in the right place. I assumed you checked what conditions those workups yield? I know that depending on the workup, you can get either the alcohol, aldehyde/ketone or carboxylic acid....so just make sure you've got the right ones.
-
Cl2 and H2 are both neutral gases. NaOH is an ionic solid and is made up of Na+ and HO-. In solution the two ions will have dissociated from each other and posses solvation shells around them. Essentially all that will happen is that you will now be trying to seperate Na and OH ions instead of Na and Cl ions.
-
The product drawn in the bottom picture is wrong...you have got two of the carbonyls in the wrong place (I won't tell you which ones as its an assigment)
-
Why do you want to remove the smell? If it has a foul smelling odor, then it'l probably be due to some of the demethylated methionine (i.e. the free thiol) as it is the thiol group that is mostly responsible for smells.
-
You do need to missing bit to make a hemiacetal as well though...its the catalyst
-
How are you going FrankenNeko?
-
He says that you are basically distilling the plastic...you melt it and the make it evapourate before your condense it again in the water. I would hazard a guess that this is not very energy efficient....you are going to need a lot of energy to melt the plastic and get it into the gas phase. In terms of that, I dont think it would be very good. However, on a larger scale (say town sized) it could work. Once problem them doesn't fix though, is getting out any impurities. There is such a HUGE number of different plastics that yo can't just melt them all down and get oil back from it...so it would probably involve alot of sorting out prior to actually using this.
-
Generate Methanol with poop and bio waste
Horza2002 replied to Kevin Keller's topic in Biochemistry and Molecular Biology
There are people working towards using biodegradable products as a source of power and raw materials. The are a few reasons why it hasnt been used yet. One of the main ones is that, you actually need a variety of bacteria to breakdown the material and then another set to convert it into useful stuff. And for the most part, these organisms don't live well together. There is research ongoing to create a strain of bacteria that can do all of the jobs, but thats still on going. -
We're not just going to give you anwsers, what specifically do you need help with?
-
Ok I will now give you some hints to one possible route, but I'm not going to give you an anwser. What do you get if you treat a carbonyl compound with a strong base (such as LDA (lithium diisopropyl amide))? Is the resulting intermediate a nucelophile/electrophile and what sort of reactions can it do? Will it do conjugate additions, eliminations, substitutions, rearrangements? Also, what type of carbonyl compound should you use here? Is treating a carboxylic acid with LDA going to be a give you what I've implied above?
-
While it is very good your thinking of a way to make the C-F bond, a lot of halogenated aryl rings are commecially avaliable. So you might want to have a look what ones are avlaiable so you roughly know what you need to do. Ok, so if you disconnect at the carboxylic acid, what are the two starting materials your going to need? Also remember, that you dont always need to break a bond, you can also use a functional group interconverstion step (i.e. like you said, make a different ester). When I do these things myself, I would now be making a list of all the ways I know how to make a carboxylic acid; what ways do you know of too make a carboyxlic acid? Then once I have all the methods, I would draw out the structures of the starting materials needed for each of those steps. Once I have all the starting materials, I would then see how to synthesis them...and then basically repeat this process until you get to commercially avaliable material.
-
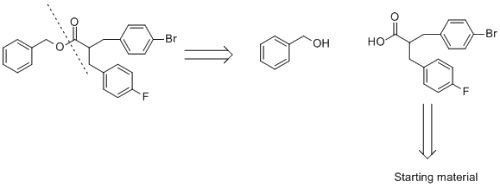
If you break an ester down, then you will get an alcohol and a carboxylic acid. You have rightly got benzyl alcohol...however your other is not correct. Firstly, H2C=C=O is not a carbxoylic acid its a ketene. A secondly the carboxylic acid fragment should contain the two benzene rings. As you knew to break the ester bond, I have shown you the first step of the retrosynthesis in the attached file...you now need to do the same thing with the carboxylic acid section. In order to do this next section, think about how you make alkyl bonds to benzene rings, or maybe some chemistry of a carbonyl type compound, maybe some substitution chemistry. You may need to do several steps in order to get commerically avalaible starting materials
-
That is where I would start yes, work backwards to see which alcohol and which carboxylic acid you need. You'll discover one of them in commerically avaliable, so thats sorted. Then you will need to break down the other half of the ester to see how you would go about making that. With a little bit more thought, breaking the ester is not actually as useful as I first thought. With that said, it does not mean that you can't do it that way... The thing with these retrosynthetic problems is if you ask 5 different chemists, they'll give you 5 different routes, with each one based on what that particular individual has mroe experience with. If you star with that ester breakage seeing as thats the one you first noticed, then have a go at breaking down the required compound.
-
As Mississippichem said, we wont give you the anwser, but we can help you get there. The idea of retro-synthesis is to look at a compound and see what bonds are easy to make and then work backwards. In your compound there, there is at least one very obvious bond that is easy to form...so I suggest you start with that one.
-
I think the best help we can offer here really is: Don't leave an assigment until the day before its due!
-
Without knowing what your compound is, it probably sounds like that your compound degraded.
-
You mean SHOULD form. As any of the organic chemists here will tell you, there is a huge difference between what happens and what should happen. Your reasoning is good, i agree that a bulky base should indeed give you the intended product. But, discrimination between a primary and secondary hydrogen is not always as clear cut as it would seem. Usually only between primary and tertiary is it clear cut. Sorry if you think I'm being picky, but I'm just trying to point out all the different angles that can occur from this
-

Synthesis of Oxalic acid from Vitamin C
Horza2002 replied to Toothpick93's topic in Organic Chemistry
I would say no, this isn't a viable route. As I said, potassium permanganate is a very very strong oxidising agent. If you exposed ascorbic acid to it, then you would oxidise the secondary alcohol to the ketone, the primary alcohol to a carboxylic acid...maybe then the carbxoyclic acids to the orthoesters....epoxidise the double bond...in short, you would get an absolutly horrible mess! -
In theory, most of that looks ok....however in practise this would not work at all; selectivly reducing one of those ketones to the alcohol is going to be extremely difficult. With that said, the step converting your dibromide to the diterminal alkenes (step two going from the start material) will not work at all. Yes in theory, you could eliminate form the primary methyl groups to give you the product you want, and maybe this will be the kinetic product...but the more substituted alkenes are more stable (i.e. four groups is more stable than 3 groups which is more stable thn 2 groups which is more stable than 1 group). This arises from hyperconjugation from the adjoining carbon-carbon or carbon-hydrogen bonds. Electron density from these bonding orbitals is able to overlap with the pi* antibonding orbital of the alkene thereofr making the system more stable. In addition to being more subsituted, your starting material is more stable as the two double bonds are in conjugation with each other.
-

Synthesis of Oxalic acid from Vitamin C
Horza2002 replied to Toothpick93's topic in Organic Chemistry
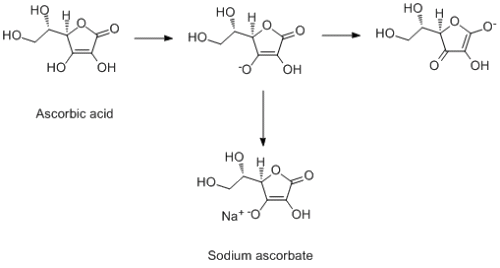
Ascorbic acid, as the name implies, is acid. This means that one of the protons is very acidic and can be deprotonated easily. All that has happened to get the sodium ascorbate, is that you have deptotonated and made the sodium salt. When you swallow the tablet, it will be reprotonated in your stomach to give you ascorbic acid back. This is an example of a tetronic acid...there is no carboxylic acid, but due to conjugation through the double bond, one fo the hydroxy groups is acidic. Now onto your synthesis bits...No, potassium permanganate is not a reducing agent, it is an oxidising agent. And the first step is actually an oxidation as well, not a reduction. A general rule, if you are increasing the number of hydrogens in a molcule, then it is a reduction...if you are dereasing the number of hydrogen, it is an oxidation (this is not 100% all the time...but for a begininger it should be ok). Potassium permanganate is rarely used as a reagent because it is such a strong oxidiser, you will oxidise absolutely everything in your molcule (that is why it used as a stin for TLC). You very rarely get hydrogen gas given off as well. Your second step is essentially an ester hydrolysis resulting in the opening of the 5-membered ring to give you the 2,5 diketogulonic acid. In theory, water could do this reaction...but in practise, sodium hydroxide is used (it is much mroe reactive). One potential problem here, is that 5-membered rings are pretty stable, and 5-memebred lactones (cyclic esters) are notoriously difficult to make them undergo hydrolysis. So this step is unlikely to work very well if at all. That final step does not seem very likely to happen at all to me...im not even sure where to begin with that one. -
It really depends on what solvent your trying to remove as well. For example, if you are trying to remove water from a sample, it is much easier to freeze dry it (freeze the sample in liquid nitrogen and then allow it to sublime under a vacuum). If your trying to remove diethyl ether...you could just leave it in a warm room for a few hours and you'd be fine.....in a fumecupboard of course.
-

How to determine toxicity based on electron configuration
Horza2002 replied to true1990's topic in Homework Help
...Sorry, but I really don't see where that question has come from given the context of the questions asked so far. Elements changing into other elements (i.e. arsenic changing into krypton) is the area of nuclear physics and radioactivity. Also, I'm not sure how easy it is for arsenic to be converted to krypton 81....natural arsenic is 100% As-75....so you need to add 6 neucleons (2 protons and 4 neutrons) to get from As-75 to Kr-81. With that said, I'm a chemist, not a nuclear physicist so maybe there are other ways it can be done