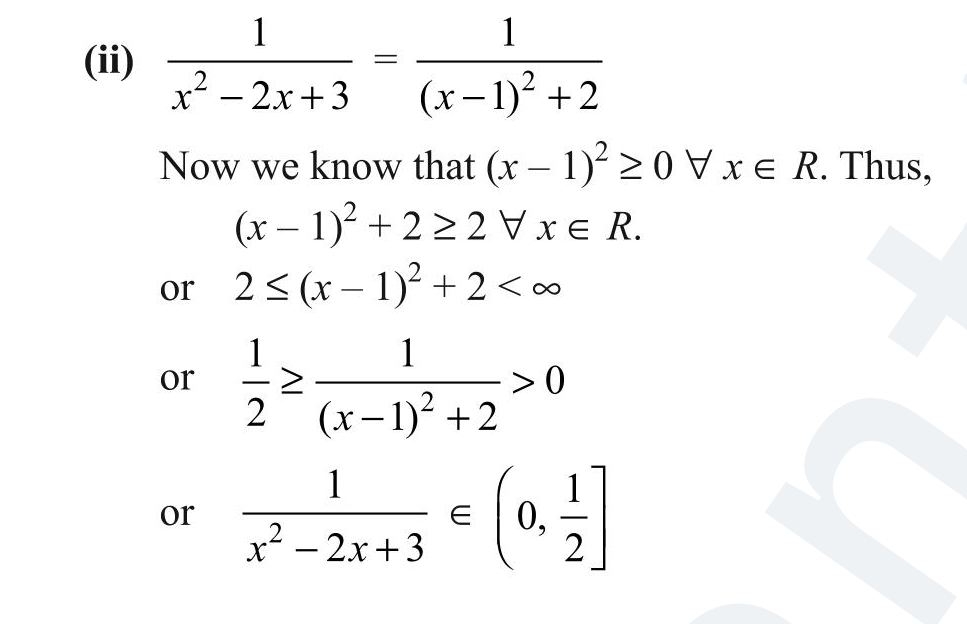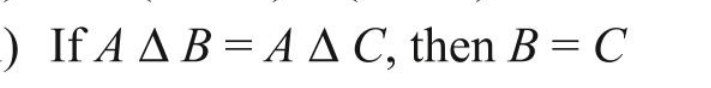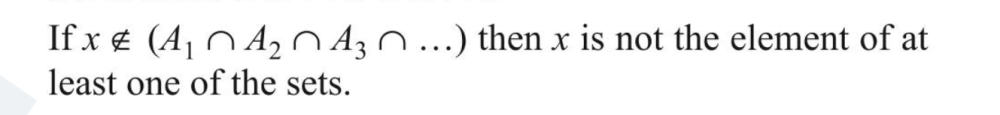Everything posted by HbWhi5F
-
≤ can mean- till it or till it OR infinity ?
equalto /greater than OR greater than /equal to Contextually I understand why it's (0,infinity) but > w/ dash under as 2 means aka till it and till it or infinity x≤1/2 can mean (0,1/2) or (1/2,infinity)
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
@Genady thanks
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
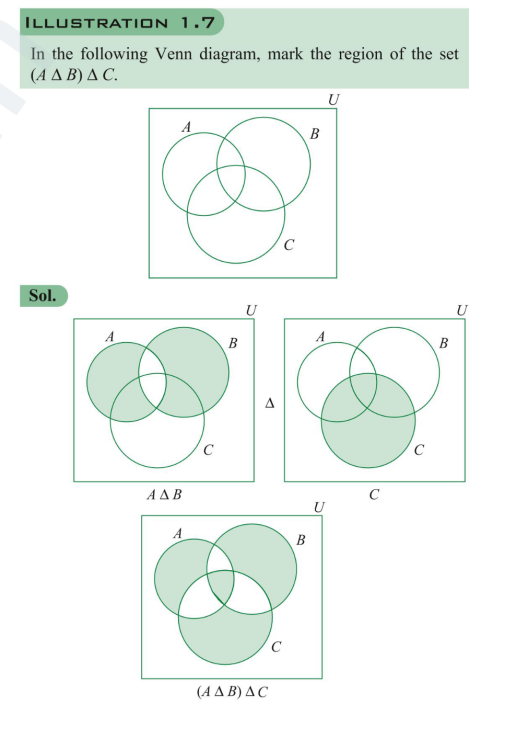
@studiot In Symmetric difference why is intersection of 3 not included, also what its associative. Symmetric difference means "unique to" ? So AΔBΔC != (AΔB)ΔC
-
when building powersets why don't we make combinations with null set ?
So conclusion is null set is a subset of everyset ands ∅ is not always a element of every set.
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
@Genady https://www.geeksforgeeks.org/maths/symmetric-difference-of-sets/
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
@Genady Explain this doesn't makes sense to me. This implies if x doesnt belong to intersection of 3 sets that means it doesn't blong to any set.
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
@Genady I am having problem understanding these
-
[Math] [Set] De Morgan's Law, Symmetric difference with 3 sets
This doesn't makes senseIf x e (A1 n A2 n 143 n ... ) then x is not the element of at least one of the sets. This implies if x doesnt belong to intersection of 3 sets that means it doesn't blong to any set. De Morgan's LawI think this is false when C⊂B with elements of A U B If A A B = A A C, then B similar to Symmetric difference with 3 setsIn Symmetric difference why is intersection of 3 not included, also what its associative. Symmetric difference means "unique to" ? So AΔBΔC != (AΔB)ΔC Cardinal Number of set Union of 4 SetsI thinkg it should be - n(AUBUCUD) = [Sum of number of elements in sets A, B, C and D] - [Sum of number of elements of intersection of sets taken 2 at a time] - [Sum of no. of elements of intersection of sets taken 3 at a time] - Elements of intersection of all sets
-
when building powersets why don't we make combinations with null set ?
It breaks the rule number of elements of a powerset of Set A = 2^n . Also contradicts null set is a subset of everyset. Also some places it says ∅ is a element of every set 2^4 = 16, but my results say 39 Example for set {1,2,3,4} There can be 5 no. of sets n+1 single element = n 2 element = 10 - {1,2} {1,3} {1,4} {2,3} {2,4} {3,4} {1,∅} {2,∅} (3,∅) (4,∅) 3 elements = 10 4 elemets = 4 =>{1,2,3,4} {1,2,3,∅} {1,2,∅,4} {1,∅,3,4} {∅,2,3,4} 5 element = 5 additional the ∅
-
[Eletrochem] Help me understand molar conducitvity
@genady i don't understand please help
-
[Eletrochem] Help me understand molar conducitvity
1 mol m–3 = 1000(L/m3) × molarity (mol/L) The 2nd equation seems to contradict the definetion, how is it using both cm and m in a forumal, i am having trouble understabding the conversion factor
-
How to better use AI for study Science ?
What I do I give it pdfs and a long instruction, and then ask questions. Best Service and Local LLMI use ChatGPT, should I switch to something else or something local ? Give AI access to screen, able Point-&-Ask while reading ? I saw video people doing similar things w/ Gemini, Claude. 1. I may use local OCR/CV and send it to LLM as text. 2. and use local Speech to text ? Should I give ChatGPT access to local files for this purposeLangChain - TechLinked - https://www.youtube.com/watch?v=9AXP7tCI9PI Local LLM Retraining, RAG, Context Docs - https://www.youtube.com/watch?v=fFgyOucIFuk How else can I use AI to study ?
-
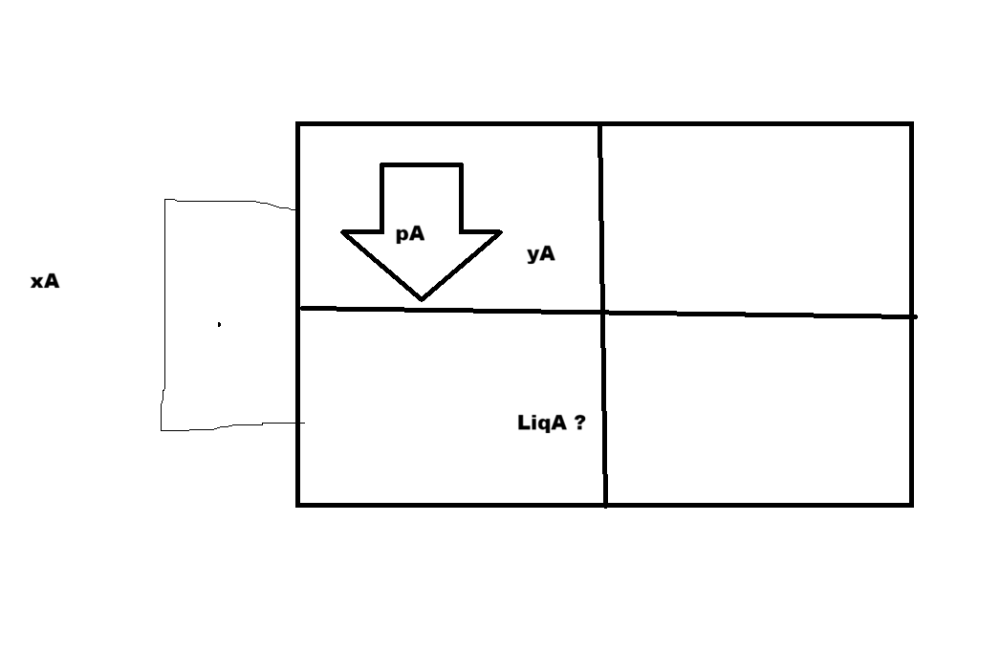
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
xA is xA/xA+XB as the question is asking liquid ratio so LiqA/LiqA+LiqB
-
[Chem-Applied-Solutions] van’t Hoff factor i
@KJW I don't get the context of "The number of moles is inversely proportional to the molar mass." in van't hoff's factor. The definition/formula in book seems self-contradictory.
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
I meant LiqA/LiqA+LiqB
-
[Chem-Applied-Solutions] van’t Hoff factor i
@KJW What is that have to do with ? I meant Normal = Calculated and Observed=Abnormal (has the definition below it says)
-
[Chem-Applied-Solutions] van’t Hoff factor i
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
I still don't know how to find comp. of liquid mix (LiqA in diagram). it is not possible to be get the value (from the infomation in the book). or I got the question wrong ? He is my guru
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
Given: p°A = 450 mm Hg (pure component's vapour pressure), °B = 700 mm Hg, = 600 mm Hg Let xA and xB are mole ractions, So xB = 1 − xA Using Raoult’s law: Total Vapor pressure = mole fraction of component A in it's vapour and liquid phase times the Vapour pressure of pure component A + same thing for B P = xA·p°A + xB·p°B 600 = 450xA + 700(1 − xA) 600 = 700 − 250xA xA = 0.40 xB = 1-.4 = 0.60 Partial vapour pressures: pA=pA^0 * xA (Parital pressure of a component = vapoyr pressure of pre component times it's mole fraction) pA = xA·p°A = 0.40 × 450 = 180 mm Hg pB = xB·p°B = 0.60 × 700 = 420 mm Hg pA and pB are vapour partial pressure, ie the pressure exerted by them on solution Vapour phase composition: pi = yi * P (partial pressure of a component = the ratio of it's vapour phase the vapour phase of the other component) times the total vapour pressure on solution yi=pi/P yA = pA / P = 180 / 600 = 0.30 yB = pB / P = 420 / 600 = 0.70
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
@exchemist updated my answer Also did i get the definations right and the question ?
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
@exchemist Given the context of the question AI is right but If I got the definattion right, it is not possible to be get the value (from the infomation in the book). Either I got the question wrong or the definetion. Ok the conceptual problem is - We got ratio amongst vapours and ratios of the components amonst their phases How are they connected ? It should be something like - Let V and L be phase of Vapour and LIquid in the solution V/L = [1/Q1] / [(xA/y1) + (xB/y2)]/Q2 Q is some ratio to normalize the value from 1 to a fraction of the solution - it should be y1+y2/xa+xb V/L=[1/(y1+y2/xa+xb)] / [(xA/y1) + (xB/y2)]/(y1+y2/xa+xb) V/L=[1*xa+xb/y1+y2] / [(xA*y2+xB+y1/y1y2) / (y1+y2/xa+xb)] 0.30+0.70/L=xA + xB 1/L = 0.40+0.60 1/L=1 L=1/1 V/L=ya/xa
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
I worked same solution but why is AI saying it's liquid phase composition ? It's -xA and xB are mole fractions of each components - Vapour-liquid phase ?
-
What is the Benzene base compound priority list ?
-
[Chem-Applied] Vapour pressure of Pure Liquids A & B are 450 & 700mmHg respect. @350 K . Find comp. of liquid mix, total Vapour pressure is 600mmHg and Vapour phase composition
xA and xB are mole fractions of each components - Vapour-liquid phase ? why is AI saying it's liquid phase composition ? Henry's Law : p = KH x “partial pressure of as in vapour phase (p) is proportional to ole fraction of gas (x) in solution” and is expressed as: Raoult’s Law: A=xA pA∘ pi = yi P total - this is the ratio of 2 components in vapour phase AI Solved - Given: p°A = 450 mm Hg p°B = 700 mm Hg Total vapour pressure, P = 600 mm Hg Let xA be the mole fraction of A in the liquid phase. Then xB = 1 − xA Using Raoult’s law: P = xA·p°A + xB·p°B 600 = 450xA + 700(1 − xA) 600 = 700 − 250xA xA = 0.40 Therefore: xA = 0.40 xB = 0.60 Partial vapour pressures: pA = xA·p°A = 0.40 × 450 = 180 mm Hg pB = xB·p°B = 0.60 × 700 = 420 mm Hg Vapour phase composition: yA = pA / P = 180 / 600 = 0.30 yB = pB / P = 420 / 600 = 0.70 Final Answer: Liquid phase composition: xA = 0.40 xB = 0.60 Vapour phase composition: yA = 0.30 yB = 0.70