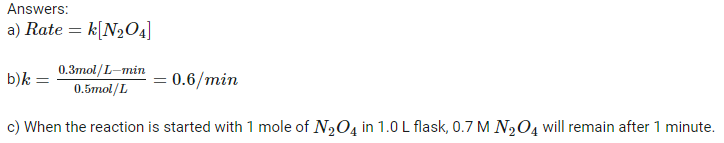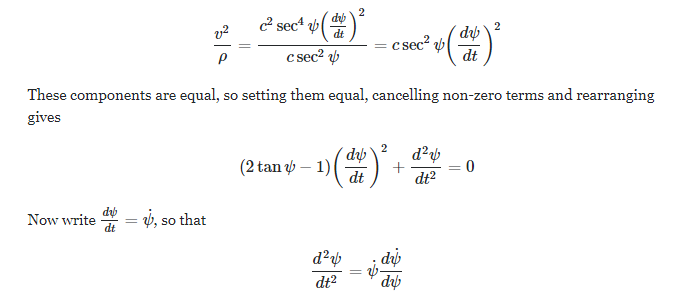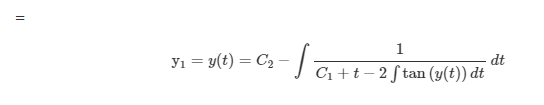Everything posted by Dhamnekar Win,odd
-
Computation of longest wavelength emission from [math]Ca^{19+}[/math]
There is typographical error in the above question. Please read ' Is [math]Ca^{19+}[/math] one electron atom? '
-
Computation of longest wavelength emission from [math]Ca^{19+}[/math]
Was the above question correctly stated and correctly answered? In [math]Ca^{19+}[/math], how many electron shells are there? answer is n=1,2,3,4 Then in ground state how can we found its valence electron in 6th shell or n=6? Is [math]Ca^{19+}[/math] one lectron atom?
-
Question on structure of Atom
Answers: REMOVED BY MODERATOR
-
Question on structure of Atom
An x-ray photoelectron spectroscopy experiment with an unidentified element, X, displays an emission spectrum with four distinct kinetic energies: [math] 5.9 \times 10^{-17} J, 2.53 \times 10^{-18} J,[/math] [math]2.59 \times 10^{-20} J, 2.67 \times 10^{-20} J [/math] (Assume the incident light has sufficient energy to eject any electron in the atom.) (a)Name all of the possible ground state atoms that could yield this spectrum. (b)Calculate the binding energy of an electron in the 2p orbital of element X if the x-rays used for the spectroscopy experiment had an energy of [math] 2.68 \times 10^{-16} J[/math] (c)Consider both the filled and unfilled orbitals of element X. Determine the number of: (1) total nodes in a 4d orbital (2)angular nodes in the [math]2p_y[/math] orbital (3)degenerate 5p orbitals How to answer all these questions?
-
How to prove the normal distribution tail inequality for large x?
How to prove (7.1)? How can we use the following two expansion formulas of CDF of normal distribution to prove (7.1) lemma?
-
Can I ask here statistics question?
Can I ask here statistics question? If yes, under which forum?
-
In Daniell cell, anode has higher reduction potential and cathode has lower reduction potential
For additional information, refer to the following table:
-
In Daniell cell, anode has higher reduction potential and cathode has lower reduction potential
- Rates of reactions [math] N_2O_4 \rightarrow 2 NO_2[/math]
Answer to d) 1)∆H = 17.4 kJ/mol 2)∆H = 8.7 kJ/mol 3)∆H= 4.35 kJ/mol Answer to e) Mechanism for this reaction is [math]1)N_2 + 2 O_2 \rightarrow 2 NO_2 \Delta H^\circ_f = 66.4 kJ/mol [/math] [math]2) N_2O_4 \rightarrow N_2 + 2 O_2 \Delta H^\circ_f = -9.16 kJ/mol [/math]- Rates of reactions [math] N_2O_4 \rightarrow 2 NO_2[/math]
I realized my mistake. The answer computed for b) is k=-0.4. But how to answer d, e ,f ,g ,h and i? I am working on all these questions. Any chemistry help from the member of this science(Chemistry) forum will be accepted.- Rates of reactions [math] N_2O_4 \rightarrow 2 NO_2[/math]
How to answer all these questions? Are these above answers correct? Answers to remaining questions will be provided very soon.- Rates of reactions questions
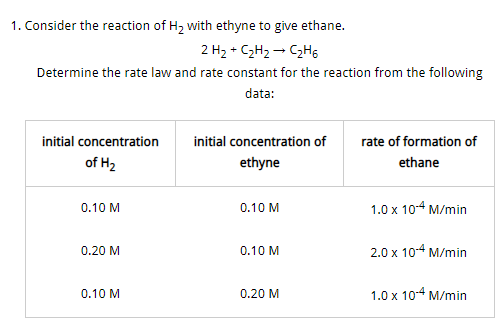
[math]2H_2+C_2H_2 \rightarrow C_2H_6[/math] is first order with respect to [math]H_2[/math] and zero order with respect to ethyne and the rate law is [math]Rate=k[H_2],k=\frac{rate}{[H2]}=\frac{1.0 \times 10^{−4}M/min}{0.1M} =1×10^{−3}min^{−1}[/math]- Velocity and acceeration [ Vector calculus with applications ]
Now, here is the final correct answer provided to me by one great expert mathematician from UK (United Kingdom). That's it.- Rate laws of some chemical reactions
- Velocity and acceeration [ Vector calculus with applications ]
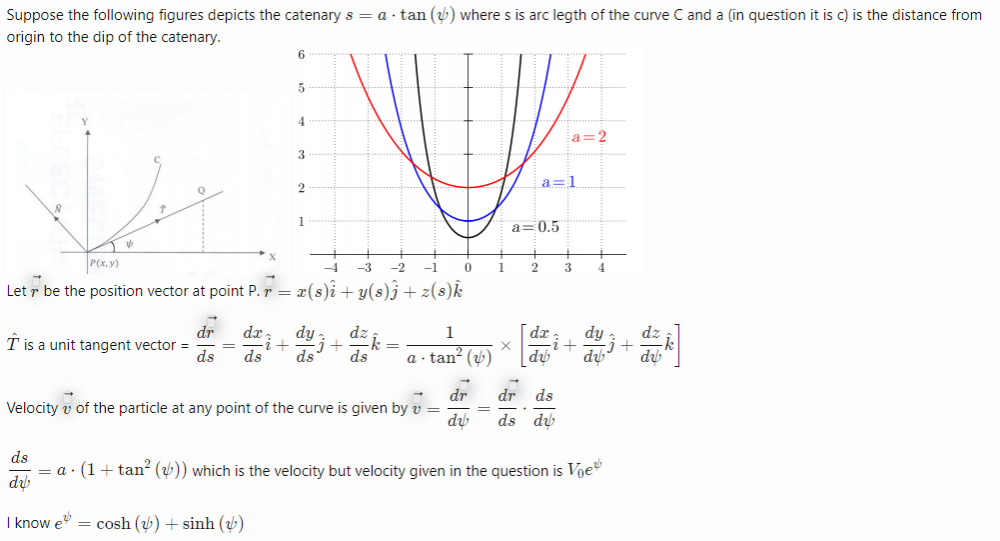
Now let us replace ψ by y and solve the aforesaid differential equation. Now we replace y by [math]\psi(t)[/math], we get, [math]\psi(t)= C_2 - \displaystyle\int {\frac{1}{C_1 + t -2 \int {\psi(t)}dt}dt}[/math] Now , how to show that velocity and acceleration of a moving particle along a catenary are equal to the given values in the question?- Velocity and acceeration [ Vector calculus with applications ]
Example: Find the radial and transverse acceleration of a particle moving in a plane curve in Polar coordinates. How can we use this example to solve our original catenary problem? We know [math] \tan{\psi}=\frac{s}{a}, s=c\cdot \sinh{\frac{X}{c}}, V_0 = c , e^\psi= \cosh{\psi} + \sinh{{\psi}}[/math] I am able to get the magnitude of velocity [math]V = c\cdot e^{\psi=0}=c,V = V_0\cdot e^{\psi}[/math] but i am not getting magnitude of acceleration [math]\|\vec{a}\|= \frac{\sqrt{2}}{c} \cdot c^2 \cdot e^{2\psi} \cdot \cos^2{\psi}[/math]- Rates of reactions questions
What is the answer to the above question? Any chemistry help will be accepted. I am working on this question.- Velocity and acceeration [ Vector calculus with applications ]
- Velocity and acceeration [ Vector calculus with applications ]
[math] s= c \cdot \tan{(\psi)}, \frac{ds}{dt} = c\cdot (\tan^2{(\psi)} + 1)[/math]. Now how to compute [math] \frac{d\vec{r}}{ds}[/math] to find velocity?- Velocity and acceeration [ Vector calculus with applications ]
s is the arc length of a particle P from a fixed point P0(S=0) on curve C. Velocity is [math] \vec{v}(t)= \frac{d\vec{r}}{ds}\times \frac{ds}{dt}[/math] where r⃗ (t) is the position of particle P at time t. Acceleration is [math] \vec{a}(t) = \frac{d\vec{v}}{dt}= \frac{d^2\vec{r}}{dt^2}[/math]- Velocity and acceeration [ Vector calculus with applications ]
- Equivalent based questions (valency factors)
Answer to d: 5 moles of Ferrous oxalate FeC2O4 are required to reduce 2 moles of Potassium Permanganate in acidic medium. Answer to e: Redox equation is [math] K_2Cr_2O_7 + 3Cu_2S + 14H^+ \rightarrow 6Cu^{2+} + 3S + 7 H_2O + 2K^+ + 2Cr [/math] From the aforesaid equation, we can say that [math] \frac56 [/math] moles of K2Cr2O7 is required to react completely with 2.5 moles of [math]Cu_2S[/math] in acidic medium.- Equivalent based questions (valency factors)
- Equivalent based questions (valency factors)
- Rates of reactions [math] N_2O_4 \rightarrow 2 NO_2[/math]
Important Information
We have placed cookies on your device to help make this website better. You can adjust your cookie settings, otherwise we'll assume you're okay to continue.
Account
Navigation
Search
Configure browser push notifications
Chrome (Android)
- Tap the lock icon next to the address bar.
- Tap Permissions → Notifications.
- Adjust your preference.
Chrome (Desktop)
- Click the padlock icon in the address bar.
- Select Site settings.
- Find Notifications and adjust your preference.
Safari (iOS 16.4+)
- Ensure the site is installed via Add to Home Screen.
- Open Settings App → Notifications.
- Find your app name and adjust your preference.
Safari (macOS)
- Go to Safari → Preferences.
- Click the Websites tab.
- Select Notifications in the sidebar.
- Find this website and adjust your preference.
Edge (Android)
- Tap the lock icon next to the address bar.
- Tap Permissions.
- Find Notifications and adjust your preference.
Edge (Desktop)
- Click the padlock icon in the address bar.
- Click Permissions for this site.
- Find Notifications and adjust your preference.
Firefox (Android)
- Go to Settings → Site permissions.
- Tap Notifications.
- Find this site in the list and adjust your preference.
Firefox (Desktop)
- Open Firefox Settings.
- Search for Notifications.
- Find this site in the list and adjust your preference.