

SirRoderick
-
Posts
3 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Posts posted by SirRoderick
-
-
Hi!
20 year old student from Belgium, currently studying for my Chemistry Bachelor at UHasselt.
P.S.
The reason I joined was to get help with this, so if you have a moment, please check it out.
0 -
In relation to a project I'm doing at university. I'm stumped on the mechanism of the reactions displayed here.
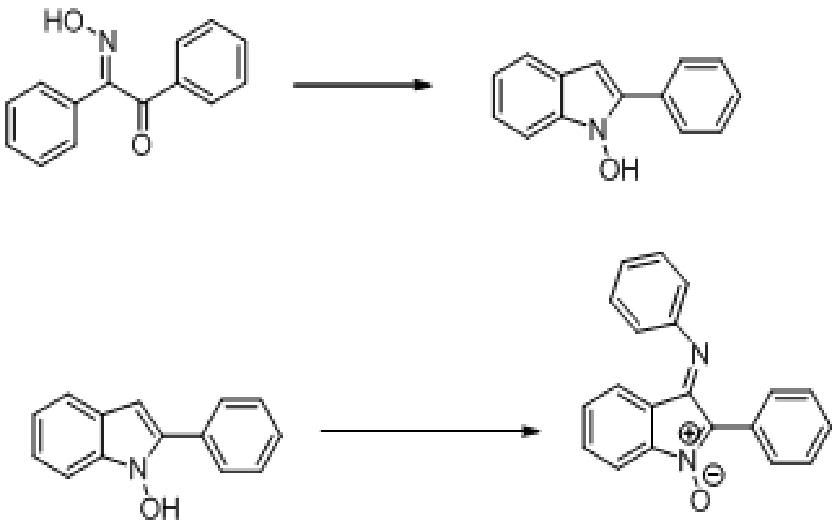
So the first is a rearrangement in sulphuric acid. Afterwards we added crushed ice (well, we add it TO crushed ice of course)
Now I don't have a scanner right here so I can't upload my sketches. But it was suggested to me that it might be easier if I flipped over the starting molecule left-to-right. (so the NOH group is on the right) He then suggested that perhaps the N forms a bond with the ring structure now to the left. This would be joined by a protonation of the O group wich would split off as water. I'm not sure this is entirely plausible.
I'm quite simply stumped on the reaction mechanism here, which is required for my presentation, well I need to at least have a plausible thought lets say.
Now as for the second reaction, it is done in presence of NaEtO in ethanol (dry), which I assume acts as a base and attacks the proton on NOH. This would then through resonance allow for the addition reaction of a nitrosobenzene. My question there is, what the hell happens to the O in the Ph-N=O that is added?
Any help would be most appreciated. I really am out of practice with this sort of thing.
0

Nitrosobenzene
in Organic Chemistry
Posted
So this is linked to my other, broader question but since I don't have any replies yet I figure I'd ask this seperately.
Nitrosobenzene, so Ph-N=O. How would the reaction mechanics work for an addition reaction to a carbon chain with a double bond? It should end up binding the N to the carbon chain and splitting off the O (presumably as water).