Everything posted by Pádraig
-
Sourcing mixing fluid online
Thanks very much to both - I'm going to purchase Glycerin on Amazon and look at formulating liquid sugar myself.
-
Sourcing mixing fluid online
(Apologies if this is the wrong forum, couldn't find one I thought suited. It's technically homework, feel free to move if there's a better fit!) Hi all, I'm looking to do some mixing at home, and measuring with a viscometer. I'll be using tap water, and one other fluid, idealy with a viscosity that's a bit higher than it. Where's the best place I could purchase a fluid that's 1) Reasonably cheap, 2) Has a viscosity at least a small bit higher than water, 3) Has its density and viscosity values available?
-
Is homogenous viscosity a good indicator for general homogeneity in mixing?
That's exactly the logic I've been using, I've just been worried that as I haven't got a high level/"expert" level of chemistry that I am/was overlooking something
-
Is homogenous viscosity a good indicator for general homogeneity in mixing?
I completely agree with this, I'd actually be tempted to shout it even louder again. I'm just saying, even in a lab situation, when crosslinking is included - does that affect the overall homogeneity, when the molecular viscosity is homogenous throughout after mixing, and why/why not? (Sorry if this seems like I'm asking the same thing again - it's just that the link for entropy of mixing gives "when several initially separate systems of different composition, each in a thermodynamic state of internal equilibrium, are mixed without chemical reaction"
-
Is homogenous viscosity a good indicator for general homogeneity in mixing?
Thanks very much - I'll look into those terms you've given me, that's a big help. Edit: @sethoflagos, this is in the second link: "mixed without chemical reaction". I'm mixing with crosslinking, will that heavily impact whether the viscous homogeneity is general homogeneity?
-
Is homogenous viscosity a good indicator for general homogeneity in mixing?
Sorry, I probably should have added - Assuming Ansys Fluent is accurate (and I am running validation tests on it), is homogenous viscosity an OK meter for finding general homogeneity in mixed fluids?
-
Is homogenous viscosity a good indicator for general homogeneity in mixing?
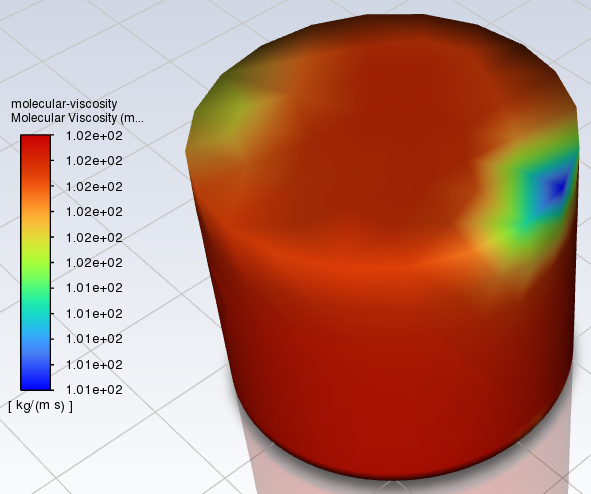
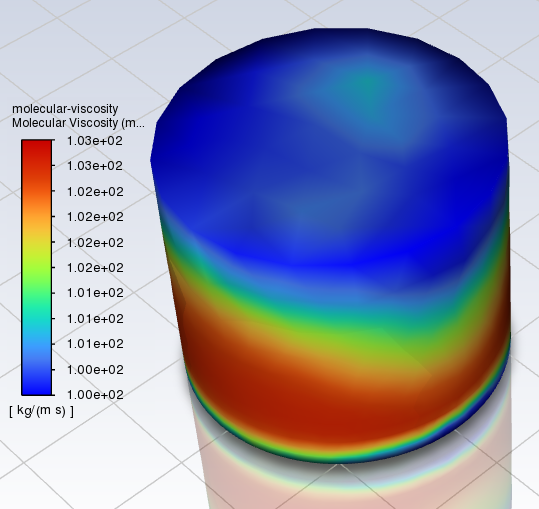
Hi all, I'm mixing two fluids, and want to find when they become homogenous. I'm using a mixing programme called Ansys, and I'm checking the mixing via the molecular viscosity. It gives this kind of result (this is after 2550 s of mixing): In this mix, I have the initial viscosities of the fluids set at 100 cSt (99 % of mix) and 500 cSt (1 % of mix) respectively. I'm mixing 60-180 s per step, until I'm at homogenous viscosity (so I can see at what time this occurs). Is this OK for finding the general homogeneity of the fluid? Here is the initial mix after 300 s, for interest:
-
Looking for fluid mixing results I can verify
I can do Static Pressure and Pressure Coefficient (both 0) and Absolute Pressure, I couldn't find any setting for Entropy
-
Looking for fluid mixing results I can verify
Hi all, I have a mixing simulation drawn up on the programme Ansys, where I am mixing two fluids. I want to verify my mixing process, by using this process to mix two other materials and take a variable of each material (e.g. viscosity) and compare the actual final mixing value to the one that I get when I mix them in my simulaiton. Is there a paper, site, or other available resource that contains mixing data for any two fluids?
-
"Unusual" result in 3D mixing
I'm getting the issue that there's no heart for Seth's OP, so when I went looking around it I couldn't give it karma; I have one for your post (both attached). Edit: reloaded page and I was able to give him the karma, heart appeared.
-
"Unusual" result in 3D mixing
That's really interesting, thank you. I've never really looked at fluid flow in that "detail" (may not be detailed to ye!), I'll look into what you've talked about now. I'm assuming I can't give you reputation as I'm a new member?
-
"Unusual" result in 3D mixing
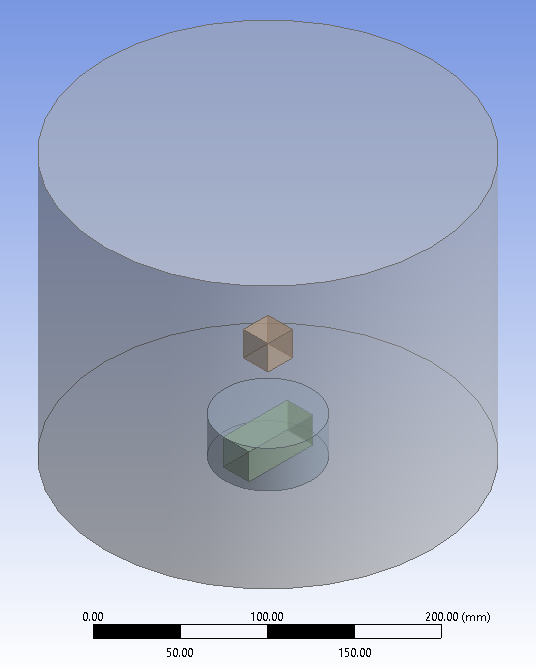
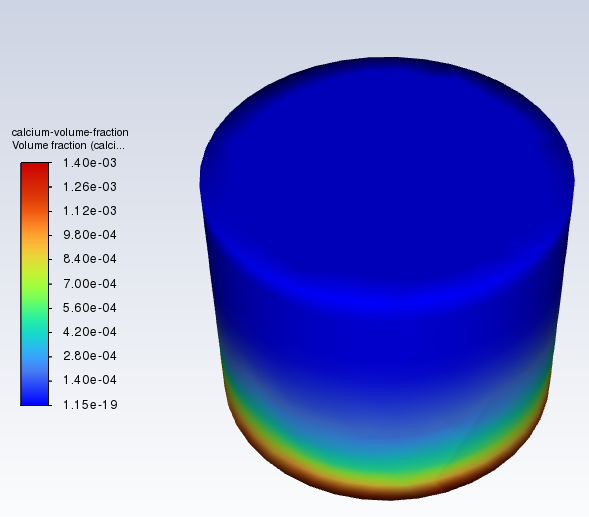
Hello, I'm currently doing 3D mixing in a computer programme called Ansys. I have a cylinder full of water, featuring a square cuboid of calcium (the rectaungular cuboid is a stirrer bar, and the smaller cylinder is a mixing zone. I have gravity defined as the standard 9.81 m/s2 downwards) : I have the mixing set at 60 rpm at the moment. I find that, after 2.5 minutes, my calcium is down around the bottom (this is a volume fraction graphic, with more blue meaning higher fraction of water, and as the colour leans into red, you have more calcium) : Then, as the mixing continues, the homogeneity of the entire cylinder stabilises much more (This is about ten minutes in. I've calculated that 3.4671e-04 throughout the cylinder is perfectly homogenous, so it's very close now) : My question (a basic mechanical/chemical one, I hope) is, why does the calcium initially "stick" to the bottom, despite being set as a cuboid a bit above the bottom at zero seconds, and then is "happy" to mix as time increases?