Everything posted by popcornfrenzy
-
Definitions
I wanna that if there is no concrete definition or widely agreed upon definition of an abstract concept then what do you do with those that are given by various philosophers throughout time. Do you choose which one to use? For example the terms Philosophy, Environment, Love, Justice, and Equality. I'm pretty sure that it doesn't work in a way like "Oh I think I'll use Plato's definition of Philosophy cause I want to," which in this case is very arbitrary? In a way, it kinda led me to the question of what is real and what is not for all these concepts which only made me more confused.
-
The Animal Kingdom
The ability to sexually reproduce (exclusive for animals and higher plants) and how they sexually reproduce maybe a factor. To think that animals are more isolated than other kingdoms especially in the terrestrial environment is a huge factor for their higher diversity. I'm also realizing a trend here, plants and bacteria have the largest biomass on our planet; while animals have the lowest according to biomass census https://faunalytics.org/the-biomass-census/#:~:text=The sum of all biomass,%2C and animals – 2 gigatons. Both the kingdom plantae and bacteria have more individuals occupying different areas in all continent; and it's interesting to note that plants also act as a habitat (with high variety), same for animals but mostly as coral reef in the marine. Plants can also act as vectors for isolation of other organisms, like in the Amazon where different microhabitats are created by plants alone. Does this mean: The higher biomass and larger land area that the organisms occupy; the lower their species diversity is?
-
The Animal Kingdom
It could also be argued that the other kingdoms have some mode of transportation that allowed them to successfully inhabit a variety of habitats. Certain genus in the bacteria kingdom like Thermus and Geobacillus are thermopile and are able to inhabit extreme environments. Same for the variety of plants, fungi, and protists that have different dispersion strategies which allowed them to be successful in other environments. There must be a collection of factors that differentiate the rate of speciation of the kingdom animalia in comparison to other kingdoms, allowing them to have a huge variety of species. A more widespread dispersion may be one of the possible factors, but still, I think there is more to it than dispersion alone. Could be, but 100-1000m average maximum dispersal of plants is no joke too, and plants have established themselves in the terrestrial landscape long before animals did. They even colonized every continent, which allowed them to adapt to multiple types of habitat. Despite that, animals still outnumbered them in number of species. Maybe it's true that the other kingdoms are not able to spread as wide as animals, but, locomotion or transportation alone cannot explain why at a hypothetically particular set of same conditions imposed in a given area in which all the kingdoms are exposed to, animals are able to speciate into higher diversity. Isolation is also a huge factor, but it can be argued that all organisms experience isolation in varying degrees but if hypothetically the isolation is same for every organisms then won't it result to same rate of speciation for everyone? I'm also curious to know that if in a simulation of a given area is populated with a single species that represents each kingdom, with enough individuals with gene variety that offsets the bottleneck effect, and they are given same amount of food or energy to consume, will the single species that represents the animals will speciate into higher diversity compared to other kingdoms overtime?
-
The Animal Kingdom
Why do this kingdom have the highest number of known species, even on estimates? I know that the kingdom bacteria (and with archaea forming "monera") alone is considered the most abundant in terms of individuals and distribution around the world but I also thought that this kingdom will have the greatest diversity. I actually event thought that plants also outnumbers animals in terms of species but apparently I was wrong. So what makes the kingdom Animalia so diverse compared to other kingdoms? This question actually popped out of my head when I searched why animals have the greatest number of species, on which the articles that showed up only describes that this kingdom has the largest number of species and not explaining "why".
-
Help in Understanding Mass Percent of Solutions in Analytical Chemistry
I actually thought that the 1 g = 1 mL is a universal thing for all solvents, glad that you guys made me realize this So basically, the activity was only for making different concentrations of NaCl, nothing more than that?
-
Help in Understanding Mass Percent of Solutions in Analytical Chemistry
How will you prepare 5% of a solution in 40 mL of water NaCl? I'm quite familiar with how this goes: 5% Nacl is equal to 5g NaCl / 100g Solution based on % by mass formula. Since 100g is equal to 100 mL The answer here would be : 40 mL H2O x 5 g NaCl / 100 mL Soln = 2 g NaCl It was stated by one of the posts in quora that max saturation of NaCl at room temperature is 357 mg / ml Link: https://www.quora.com/How-will-you-prepare-5-of-a-solution-in-40-ml-of-water-NaCl I had a practical exam similar to this and I horribly failed because I thought the 15% NaCl which around 36 grams of NaCl (I think, I already forgot, but this is only for context) will be deducted to 250 g H2O; therefore, I mixed 36 g NaCl and 214 g H2O in a volumetric flask to make a 250 g solution. And since 1 g = 1 ml, I made an assumption that my solution will be exactly at 250 mL too. I was so wrong, and the solution barely reached the line in the volumetric flask. I was also expecting that the resulting solution will be at max saturation, thinking that saturation was the point of the activity. Anyways my question is, do these kind of problem actually takes into account max saturation? Or does it only help in preparing an specific % of solute to solution ratio regardless whether the result will be unsaturated, saturated, or supersaturated? Follow up: What is the point of this activity? Like if ever that this is not about saturation then why do I have to make random concentrations base on percentage? Shouldn't a standard solution should be followed at fixed percentage? Like I would rather have a saturated solution (which already have tables in fixed values in the internet at room temp) for experiments than like use 10% NaCl, 15% NaCl or 50% NaCl.
-
Help for chemistry assignment
I'm currently in my third year as an environmental science student, this is for my subject in Analytical Chemistry. Actually for industrial, and domestic uses, the name muriatic acid is still used in my country, Philippines. We were told to check the labels on the internet in search for common concentrations of these chemicals. Does this mean that regardless if its w/w or v/v; the equation you mentioned is still applicable? Thanks for the reply
-
Help for chemistry assignment
How do you calculate the % water ? Let's say the % purity of Muriatic Acid is 18% how do you calculate the % Water? In terms of the reason for difference what should I look at? Like the dissolution? IDK how I would compare these solutions as they have different uses and properties.
-
Research Design
Thanks for the answers, I'll look into these. I am just worried that if I ever formulate my research questions, I'll struggle a lot with finding the right methodology to use. It sort of bugs me that I do not know these things at the same time, thinking of having a carefully structured paper to pass my course subject.
-
Research Design
Hi CharonY, in a case that there are aquifers that I can trace up and down stream of the mine, how will I determine my control? Wouldn't the other aquifers be contaminated as well? These are just my assumptions. Also, to be clear, are monitoring/surveillance studies not experimental? Even if for example you isolate the heavy metals in the lab to examine their concentrations? If not, what are they classified to? Oh, that clears the biodiversity aspect. But what if you're doing a taxonomic study as baseline for species composition in a given ecosystem or area? Like using only dichotomous keys. Because I've read some papers that did those kind of methodology and they still confuse me on what research design do they fall into or if they are even quantitative at all. For example this paper: https://www.researchgate.net/publication/327654431_Freshwater_microcrustaceans_Cladocera_Anomopoda_and_Ctenopoda_Copepoda_Cyclopoida_and_Calanoida_in_the_highly_urbanized_Metropolitan_Manila_area_Luzon_Philippines The study is about the microcrustaceans that were identified in Manila using different dichotomous keys. Are these kind of studies experimental? If not, in what design do they fall into? Btw, thank you for clearing up the first topic
-
Research Design
I'm currently working on research proposals and I can't seem to wrap my head around the idea of what research design my topics falls to. Topics: Identifying zooplankton biodiversity and its effect on the quality of local fisheries of grouper fish - I was told by my tutor that it is a quasi-experimental design but I don't get how it is since there is no manipulation of the variables since I'm just identifying the zooplankton present in the fisheries and looking if the groupers are affected in terms of their physical attributes, etc. I think it is more of a correlational study? Still, I need a clearer viewpoint on this. Analysis of the presence of heavy metals (specifically copper) in the local groundwater sources in a nearby abandoned copper mine- I also don't know if it is a experimental research since it also does not manipulate the variables and merely examining the properties of the groundwater sources. I also wanna know what data analysis suitable here? Like should it by like a significant difference thing? Like a comparison between the ideal/natural concentration vs the contaminated area? The more I look at it the more it resembles a controlled and treatment like experimental study. However it still sort of natural or observational since I didn't manipulate the presence of heavy metals in the area and it didn't occur in a controlled environment like in a lab. I'm actually confused. Identifying biodiversity - In general, is it experimental if I'm using dichotomous keys to identify species composition in a given ecosystem? If so, how do you do a statistical analysis for this? Thank you in advance for the responses 🙂
-
Help with calculating the number of milligrams present in the solution
Okay noted Sir I'll try to solve it with the best of my ability thank you for the example
-
Help with calculating the number of milligrams present in the solution
That's the full statement of the problem Sir Studiot and I really don't know what to do at this point. I kinda think it's more a given rather than a problem.
-
Help with calculating the number of milligrams present in the solution
The problem: A liter of a mixture pf 0.4 Sodium Chloride and 0.2M Potassium Chloride. I really don't know what to do with the compounds present in the solution. Do I need to add them? I'm really lost. This is the last problem in the activity, the others are easy to understand and solve, but for me this one is hard to comprehend. Maybe I need to calculate for their masses individually? What do you guys think?
-
Help with determining the minerals on a sample rock
What our activity want us to identify: list the number of different minerals in the sample and give a description of each one. It is a 4 items activity, I didn't attach the others since they are easy to identify. Please help me with this one.
-
Help with determining the least dangerous volcanic landform
I'm sorry for the misunderstanding I thought the ones you mentioned sir are all dangerous. I think our instructor is referring to now when he gave us this question because it concerns humanity. Honestly I never thought that this post will blow up but now I think I'll go with what sir @exchemist stated since it was never mentioned in my textbook that volcanic necks are indeed dangerous. Thank you all for the answers you gave to me I appreciate every information that I get from these replies.
-
Help with determining the least dangerous volcanic landform
Thanks for the answer Sir oh I really thought that it was actually stratovolcanoes that are dangerous since they've taken a lot causalities in the past. For the volcanic neck I found out in wikipedia this passage. "When present, a plug can cause an extreme build-up of pressure if rising volatile-charged magma is trapped beneath it, and this can sometimes lead to an explosive eruption." Sir umm I just want to further verify which is really the most dangerous and which is the safest, sorry for validating too much. Sorry for taking some of your valuable time here guys I'm really grateful for the help you are all giving me
-
Help with determining the least dangerous volcanic landform
Thank you sir, you're right I'm actually taking the second semester of my freshmen year. One of our subjects is geology and geography here we kinda tackle in depth how geological processes occur on earth. In my question I was actually asking for the least dangerous among the landforms I mentioned, the most dangerous one, in general are mentioned to be the stratovolcanoes so I kinda relied on that. In terms of what I was looking for, I can't seem to determine which is the one that will be considered "safe" though I kinda doubt that word after researching a bunch of stuff in the internet.
-
Help with determining the least dangerous volcanic landform
1. If you were to rank volcanic landforms in terms of the risk they pose to humans, which would be the most risky landform to encounter, and which would be relatively safe? Explain your answer. This is the question in my assignment, I was able to determine the the most dangerous, which are the stratovolcanoes though research, but when I was determining which is the safe one I've encountered a lot of problems. Here are the choices base on my textbook - shield volcanoes, cinder cones, calderas, fissures, basalt plateaus, lava domes, and volcanic necks. Based on what I've searched on the internet, all of the the volcanoes mentioned are dangerous, the calderas are still dangerous due to the active volcanoes that are commonly beneath them like in the case of the yellowstone volcano, fissures are dangerous and can lead to basalt flow or plateaus that an article mentioned to be more dangerous than a super volcano, lava domes can also collapse and create destruction, and lastly volcanic necks can explode due to the buildup of pressure beneath them. At this point I don't even know what's safe anymore.
-
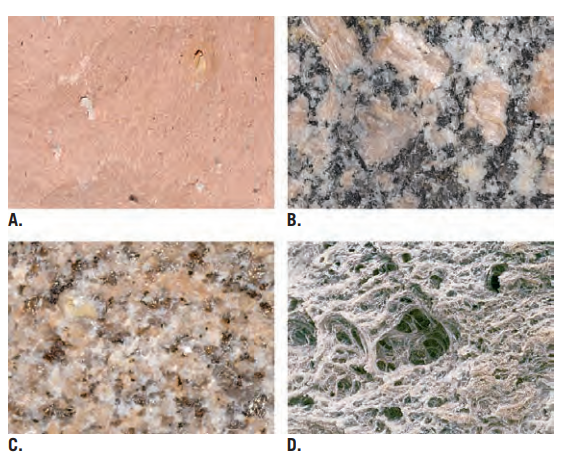
Help with identifying igneous texture
Aphanitic: (Pink) Rhyolite Porphyritic: Andesite Phaneritic: Diorite Granidiorite Granite Granular: Quartzite I kinda forgot the fact that coarse-grain is the only texture that forms within the Earth in contrast to the others that are formed on the Earth's surface. I actually think that C is indeed Granite based on this picture I found in the internet. Thanks for the comparison I think I know now perhaps? I'm open for validation Sir
-
Help with identifying igneous texture
I managed to identify A B and D what got me confused is the texture of C I kinda think it's Phaneritic based on how similar it looks to B in terms of the texture, but I've searched things related to the picture on the internet and seen people saying it's Porphyritic. So what do you think guys? Plus knowing what kind of rock it is will also help, though I do think it's a granite, correct me if I'm wrong thank you.
-
Nature of Magma
Oh I'll take note of that isn't the SiO2 the empirical formula or did I get that wrong?
-
Nature of Magma
Thanks to both of you for the answers I can finally get a clearer picture on how these elements behave as a compound in the magma, I actually thought oxygen exists here as an ion along with the others that I mentioned. Good thing it was cleared up.
-
Nature of Magma
I just want to ask how oxygen, silicon, aluminum, potassium, calcium, etc., basically the liquid portion of magma, how are they in liquid form? In our textbook, it was mentioned that the melt consists of mobile ions of these elements so does it mean the liquid part is a congregate of ions in the form of ionic liquid? The part that got me confused is with the characteristic of oxygen because it only exists as a liquid in -183 oC, presumably at lower temperatures, while the asthenosphere is around 1300 oC which makes me think that it should be in a gaseous form. Why is it a liquid along with the other elements I mentioned?
-
Redox and Electrochemistry
Oh thanks I realized a lot things especially with the addition of salt and sulfuric acid though I'm understanding the concept bit by bit gladly I'm on the right path