

id86
-
Posts
2 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Posts posted by id86
-
-
Hi,
I wonder if someone could help. Im currently an undergraduate student and my organic chemistry is not good and I would like some advice with a reaction mechanism.
I am trying to work out the reaction for the cyclisation of creatine to creatinine in acidic conditions. All I have been able to find online is the first image.
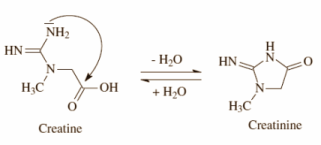
My apologies of there is nothing else too this and that is all there is too it.
I have been trying for a while to figure this out, one pathway I have attached below but I can see this is wrong but its the closest I can seem to figure out the would depend on the H+ ions in acidic solution.
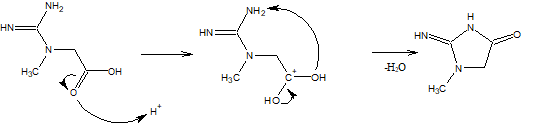
I know i'm probably completely wrong and sorry if this is a stupid question.
I believe the first reaction probably is all there is as I cant seem to make sense of anything further, and my problem is I do seem to overthink things and over complicate matters.
But what I am struggling to understand is why the reaction occurs faster under acidic conditions if the free H+ in solution do not react, why does a decrease in pH make this reaction occur faster. Also under acidic conditions, would the oxygen on the carbonyl group not also react? why would the reaction stop there?
Any help/advice would be appreciated
Thank you
0

Creatine - creatinine conversion
in Organic Chemistry
Posted
Thank you so much. That makes sense.
I think I may have made a mistake with the question regarding why it occurs faster. I understand that the addition of the proton to the carbonyl oxygen creates a dipole allowing the amine group to bond. What I've read says that a lower pH acidic solution will make the reaction occur faster. so pH 3 will be faster than pH 4. If only the one H+is being used from the acid solution, why will an increase in free H+ ions make that occur faster, unless it is down to the fact more molecules have the potential to be protonated faster or dehydration step can occur faster?
Again I do apologise if this seems obvious. and thank you once again for your help.