

tareek
-
Posts
3 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Posts posted by tareek
-
-
1 hour ago, exchemist said:
Carboxylate anions are not very strongly electrophilic, if I remember correctly. What do you think will happen?
Something has gone wrong with your post.
I dont know i would say the benzoic acid, will be formed back. But are you sure Acetophenon can't forn
0 -

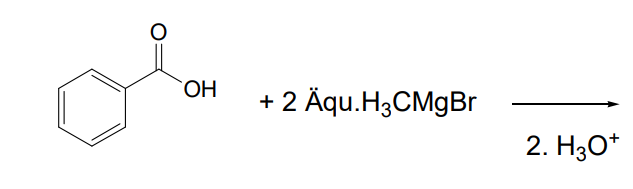
Grignard with benzoic acid
in Organic Chemistry
Posted
Because we only have 2 equivalents of Grignard