

JessicaP
-
Posts
4 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Posts posted by JessicaP
-
-
2 minutes ago, studiot said:
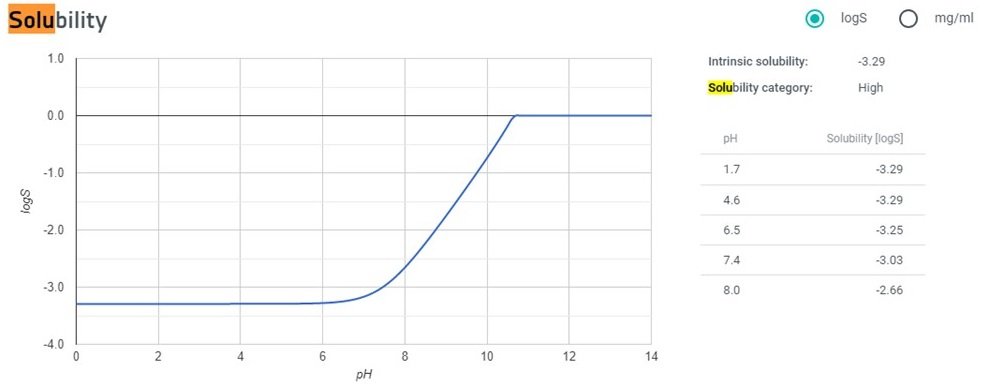
Thank you for your time. I need to understand the solubility in water. I saw that the logP is 2.03 and thus my molecule is not soluble in water. However the solubility graph on chemicalize tells the opposite.
 0
0 -
10 hours ago, hypervalent_iodine said:
A brief look suggests that the solubility isn’t actually that high at all, about 0.55 g/L, but this would be pH dependant and I’m not sure if my value was of the neutral form or not.
I also saw that value of solubility. The graph on chemicalize remains a mistery to me.
Thank you for your answer!
0 -
Secobarbital has a logP value = 2.03 an so is not soluble in water. However, looking at the logS/pH graph on chemicalize, it is marked "Solubility category: High".
Thank you in advance.0

What is the difference between logP and logS regarding solubility in water?
in Organic Chemistry
Posted
Thank you!