-
Posts
378 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Posts posted by ChemSiddiqui
-
-
When naming chemicals, “an” stand for c-c bonds and “en” stands c=c double bonds. For example, Propene means 3 carbon atoms consisting of double bonds within the structure
Prop – 3 carbon atoms
en - double bond
I saw in an article, the following name with the structure –
cyclohexane : CH2 - CH2- CH2 -CH2 -CH2- CH2
“an” means no double bonds. Doesn’t the above structure have double bonds ? shouldn’t it be cyclohexene ?
First of all dont take that for granted, there are rules of nomenclautre which you must follow in order to name organic compounds. As for your question, the best thing will be to draw the strucuture and see if the structure is right with 'single' bonds? Here is the structure;
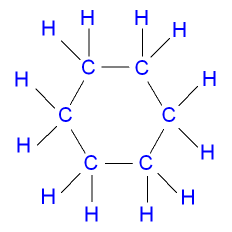
now from the stucture every carbon is tetravalent, so the formula is correct and the name is indeed cyclohexane.
For cycolhexene, there is one unsaturated bond present in the molecules , not 'Bonds' as you suggested. the sturucture is;
the forumula of that would be ;
CH2-CH2-CH2-CH2-CH=CH
hope that helped!
0 -
I wonder why you are given questions about 3D particle-in-a-box when it seems you haven't even discussed the 1D case, yet. I'll see if I can construct a short walkthrough for the problem but that'll take a few hours (during which I should better not fall asleep). EDIT: Won't get that done today; will update sometime on the weekend.
well we have been given a couple of lectures on 1-D box; this is what i gathered from them;
when an particle (say electron) is trapped in a box of one-dimension (hence particle in 1-D) of legth x1 =0 and x2=L and it can translate freely along one direction only say x-direction then the probability of finding the electron between the two ends of the box is given by the born interpretation of the wavefunction. also the probability of finding an electron at the walls is zero as wavefunction is zero(potential energy at the walls is infinity) and also outside the box the probability of finding electron is aslo zero. The TI-schrodinger's equation can be formed by solving the hamiltonian operator.
H = E + V (note the sympol '^' is over each letter in the formula).
E is the kinectic energy which is by the virtue of electron transition and V is the potential energy which is due to interaction. Because there is just one particle and it cant interact with anyother particle the potential energy is zero.
So the hamiltonian opertor is H =E and schrodinger's equation can be written as;
H(psi) = E(psi)
- h(bar)^2/2m * d^2(psi)/dx^2 = E(psi)
although we have not been taught to solve the schrodinger's equation however we have been told how to 'keep' the acceptable solutions and 'discard' the unacceptable ones. This is as far as we went. And like I said in my previous message 3-D will be done in more detail next year, we have just for the time being told that for the 3-D we have to multiply the solution to 1-D by 3. but I am not sure if its the same for energy? O yeh, we also covered sketching wavefunctions of 1-D box!. And Thanks a lot for helping!
0 -
No I think you fundamentally messed it up.
Fine till here except that
- I think n=3 would also be fine (or why not?),
- the question does not ask for a particle in 1D,
- you should ask yourself what happens for n=0,
- I did not check the numbers or the term for the energy, only the algebra.
Where do you get that from? The degeneracy for a 1D particle in a box without additional degrees of freedom is 1 for all energies.
You should look up the 2-dimensional particle in a box and understand things like [math]H = H_x + H_y \Rightarrow \psi(x,y) = \psi_x(x) \psi_y(y)[/math] (H being the hamiltonian, psi being the time-independent wave-function and x and y being the respective coordinates) and what that means for the quantum numbers (particularly their number) and the total energy and what [math]\psi_x(x)[/math] and [math]\psi_y(y)[/math] are.
???
the question asked the degeneracies of states with energy lower than the given one. n=3 is the state with the given energy so lower ones should be n=1,2, right? Also, since its my first year in university and we only been introduced to QM. We have been told that we could use particle in 1-D box model for particle in 2-D or 3-D or N-D boxes. all we have to do is to times the solution of the 1-D model by the appropriate number of dimensions. About the degenericies I think I looked it up from the hydrogen atom spectrum. I understand the hamiltonian operator concept, The kinetic energy is equal to
-h(bar)^2/2m * d^2(psi)/dx^2.
The Kinetic energy is 0 because of no interaction with any other particle (infact there is one one electron inside the box). but I am still not sure about the question. Could you tell me where I was wrong. thanks
0 -
Ok here is one question I did, but I am not sure of answer. I am asking questions about QM because I am new to this subject area. Here goes the question;
An electron is trapped in a three-dimensional box with edge lengths Lx=Ly=Lz= 6.626 x 10^(-9) m. Among the energy levels lying below
E = 1.35 x10^(-20) J, how many are degenerate? What is the degeneracy of each one of them?
Now this is what I did.
the energy of electron in a 1-D box is;
En = n^2 *h^2/ 8*m*L^2
inputing the values to get n
n = ((En*8*m*(L)^2)/(h^2))^1/2
n =(9.83)^1/2 = approx. 3
So the energy level that lie below the given energy are n =1,2. And the degenercies are 1 for n =1 and 4 for n=2. Degeneries calculated from n^2.
Is this calculation and result right. Could someone please verify? Sorry had to use excel format for calculation! Thanks a lot for any help!
0 -
not sure about the first question but the second answer is easy if you simplify your answer to qualitative:
a catalyst increases the rate of both the forward and reverse reactions
I could give the qualitative answer but its physical chemistry and I have to expalin it quantiatively, not that the question says so but becuase physical chemistry is about that after all isn't?
Actually with a bit of reading from a text book I didnt read before, I now know that even though pressure doesnt affect the K, but equillibrium composition does change.
thanks for the answer anyway!
0 -
Ok need help with a couple of questions;
I am doing past papers and there is this question which I have attempted on my own but am not sure if I did right.
A sample of hydrogen gas is held in a pressurised reaction vessel at 350 K and 2 bar claculate ;
(i). the mean speed c(bar) (ii). the collision frequency z
It also has given the sigma(H2) =0.27 nm^2
(i) I used the mean speed formula (8RT/pi*M)^1/2 to get 1924.83 m/s. The only thing I am not sure about is if I can use this formula(the confusing thing for me is the pressure of 2 bar, not standard pressure).
I can calculate z, once i know that i calculated the mean speed right. Can anyone tell me if i did?
Second question was to explain why catalyst doesn't alter the position of equillibrium for a chemical reaction.
What I think is that catalyst doesnt change the thermodyanmic constant, so that the free energy isnt affected. The relation b/w free energy and thermodynamic constant is ;
delta G =-RTlnK
again i am slightly confused because the pressure doesnt affect the thermodynamic constant also.
Any help please.
0 -
I was wondering if it is possible to tell if an object's physical property will exhibit a quantum effect? Can we make some sort of measurement to check?
Taken from Wikipedia (http://en.wikipedia.org/wiki/Matter_wave);
his hypothesis would hold true for both electrons and for everyday objects. In de Broglie's equation an electron's wavelength will be a function of Planck's constant (6.626×10−34 joule-seconds) divided by the object's momentum (nonrelativistically, its mass multiplied by its velocity). When this momentum is very large (relative to Planck's constant), then an object's wavelength is very small. This is the case with every-day objects, such as a person. Given the enormous momentum of a person compared with the very tiny Planck constant, the wavelength of a person would be so small (on the order of 10−35 meter or smaller) as to be undetectable by any current measurement tools. On the other hand, many small particles (such as typical electrons in everyday materials) have a very low momentum compared to macroscopic objects. In this case, the de Broglie wavelength may be large enough that the particle's wave-like nature gives observable effects.
So could we say that measuring the de broglie wavelength can tell us if the objects physical property will exhibit a quantum effect?
thanks in advance for any additional help.
0 -
well pipette might be of some use but you wont know the titre volume unless the end-point reaches exactly when you have emptied the pipette of the HCl. Also, burette are the most accurate glassware for titration.
0 -
1. Orbitals in many-electron atoms are lower in energy than the hydrogenic orbital.
i think they are talking about corresponding orbitals of hydrogen and other elements. here due to higer charge on nucleus will result in higher attraction hence lower energy. ie 1s e- of other elements will de lower than the 1se- of H similarly 2s,2p... of others will be of lower energy then that of hydrogen.
How so?
I think from the equation we could solve this problem quantitaively,
(schrodinger wave equation cant account for energies due to e-e repulsion);
E=- kZ^2/n^2 which describes the dependence of the energy with the principal quantum number. but I cant think of a qualitative answer to this except shielding and penetration. Does 2s or 2p penetrate the nucleus better than the 1s. the radial distribution graph for the 3 orbitals seem to suggests so. If that is, problem solved!
0 -
hi all,
just a couple of points I want clarification on;
1. Orbitals in many-electron atoms are lower in energy than the hydrogenic orbital.
I dont understand this bit. when the principal quantum number increases, the radial function increases, so the energy should infact increases due to increased screening effect. But this statement says the opposite!
2. d orbitals are poorly shielded so that nd orbital is of slightly higher energy than (n+1)s orbital for a gasesous element. e.g. ...4s<3d...!
I dont get it, when d orbitals are poorly shielded they should be penetrating the core much better than (n+1)s orbital and experience the Zeff more and thus should be lower in energy.
can anyone explain these two points please or maybe direct me someplace where I can find the answer.
thnx
0 -
I'm not sure if you'd consider any of those things to be "complexes" per-se, which usually refers to transition metals with bound ligands. Perhaps the right word for what you want is a database of crystal structures? Maybe someone in the geology department (if you have one) could help if the chem. department is useless.
nicely pointed out thanks, my mistake .it was just written down in haste, i think. I dont need them anymore because I wanted them to be put into slides i was making but now they arent needed.
thanks for your suggestion though!
0 -
does anyone know where to look up the structure of complexes.... I am after strontium carbonate , sodium nitrate and diaqua calcium sulphate.
Any link that would lead to answer to my question will be great. Thanks!.
0 -
lecture was cancelled so came back home pretty soon. Had to walk all the way back (its 20 minutes walk from university), got soaked in rain and now I am doing revision for exams which are only 3 weeks away...bad times!
0 -
Who would have one what? One ice cream?
In the end, Rocky wins.
Please add an option "I just felt like voting" next time, for people like me... I just like to click teh buttonz
Now thats a funny way to put ones mistake:doh:! who did u vote anyway?
0 -
Ok I was just wondering if Muhammad Ali (G.O.A.T) ever fought Rocky Marciano (The Brockton Blockbuster), both at their peaks, who would have come out victorious?
Muhammad Ali Career:
Total fights 61
Wins 56
Wins by KO 37
Losses 5
Draws 0
No contests 0 (Source: Wikipedia)
Rocky Marciano Career:
Total fights 49
Wins 49
Wins by KO 43
Losses 0
Draws 0
No contests 0 (Source: Wikipedia)
I personally think that Ali would have owned Marciano, He was fast, he was ring intellegent and he would have psyched out marciano before he had even entered the ring with him. Rocky was of course great but I think Ali was/is the Greatest of all time.
Lets see what you think!
0 -
Pd?
in The Lounge
I brokes my glasses recently and I want to buy a new pair. I have the prescription and everything...everything except the PD which I believe the optician didn't mention in my prescription maybe because he thought I might buy specs from him. Ha, but I got them made anyway as the standard PD is 63mm something, but now that i broke the glasses I want to get my exact PD?
I was wondering if you can get it from an optician without having to take the eye test and can we get it for free. Anyone have had similar experience? I live in the Uk so will be interested to hear from usesr from there please.
thanks.
ChemSiddiqui.
0 -
j d lee, well its not the book we are recommended here in the Uk, usually its housecroft and Shrape which is used in most universities. I will try to work out the answer to it myself and from the points you mentioned up there. I did consider some of the points but hopefully I will crack it up fully!. Thanks for your help amit.
0 -
jahn taller effect is usually used in explaining diff. bw octahedral and square planer complexes. field splitting (and hence stability) is always greater in octa hedral complexes as tetra hedral complexes have field splitting=4/9 of octahedral.
hence tetra hedral are formed :
1) when ligand is weak so dufference in splitting is not that much.
2)tetra hedral arrangements are symmetrical.
have u consulted any text book? its a question came recently in my university exam. i can write whole ans. but wont it be against sfn's policies of only giving hints
fair enough, I am also not looking for the answer!. I just want directions to lead to the answer. I have an inoragnic text but that isnt very useful as far as this question goes. I tried to google it but with no avail. I totally understand your not revealing the answer and i would aslo advise you not to cause i too wouldnt go against the rules. Thanks though!
0 -
I am not too sure about what you said there. With cft we will have to assume that the ligands are point charges and as I am sure you will know it not strictly true. Also, I fail to see it having to do anything with geometry because I want to know why Co+2 mostly forms tetrahedral complex while Fe+2 forms octahedral, as both of them have rougly the same electronic configuration.
I think I might have to take into account John-Teller distortion?
what u think!
0 -
substract 80 from the M+ peak, you get 151-80=71. Now what you got to do is put on your thinking cap and manipulate between different atoms(which are involved in paracetamol) to arrive at the right fragment.
its quite basic and i assume if you have generated the mass spectrum yourself you already know this but thats all i can help now.
0 -
Hi,
I was just wondering why Fe+2 will usually form complexes which have an octahedral geometry while Co+2 will form a tetrahedral complexes. Is there a way from which we can tell that a following metal ion will form what type of complex? Can we use the Valence bond theory to do it?
0 -
hi all,
I was just wondering if the d6 ions can have spin-only magnetic moment in a tetrahedral field which gives the value 2.83 BM? I cant see that happeing becuase in tetrahedral field the spin is HIGH, and we have 4 unpaired electron to give 4.90. We dont observe the low spin in which case the spin-only magnetic moment will indeed be 2.83.
I am confused, I have got a question which asks why d6 ions in tetrahedral field have the magnetic moment 4.90 BM or 2.83 BM while 4.90 BM or 0 BM in an octahedral field. But it does ask which one of the 4 values are not observed in practice!
0 -
well for kids Harry potter beats lord of the rings hands down. but i dont no i like the magic and the story of harry potter
HP is well advertised because after a long time a british author has succeeded in attracting foreign audience. I dont know J.K.Rowling in my opinion created a novel which circulated around a pillock who as rightly pointed out here has no sense of real magic, is plain stupid and who just make use of his friends help to look like a hero. I cant see why children are so obsessed by HP; its got a similar plot to LOTR (dark lord rising and a group of people going against him etc), its characters arent that cool and its got a touch of romance which i dont see fits really well in a fantasy novel.
I might be biased but LOTR is WAY better than HP.
0 -
Man goes for a walk around Dublin. Nothing happens.
What is that all,
 ! now thats hilarious how/why would someone write a novel 1000 pages worth and nothing happens!. is it so or you are being biased?. I dont know i read a bit about this novel on wikipedia and from what i read it will be a very boring novel....no disrespect to bascule's taste. I guess I wont buy it then and no its not your opinion that made my mind up!.0
! now thats hilarious how/why would someone write a novel 1000 pages worth and nothing happens!. is it so or you are being biased?. I dont know i read a bit about this novel on wikipedia and from what i read it will be a very boring novel....no disrespect to bascule's taste. I guess I wont buy it then and no its not your opinion that made my mind up!.0


Am I doing right?
in Quantum Theory
Posted
Thank you for your post, its great and helped a lot. Just before you post the other stuff could you check this please;
The question as I undestand it is asking to find the value of n given the energy and the dimension in the three directions (x,y,z). I think we could only use the hamiltonian operator for 1-D box because the dimensions are equal i.e. Lx = Ly =Lz = 6.626 x 10^(-9), so they are dependent of each other. If they were independent, that is to say, their dimensions were diffenent Lx is not = Ly is not = Lz then we could we the hamiltonian operator for the 3-D box with the energy as follows;
Enx,ny,ny = h^2/8m ( (nx/Lx)^2 + (ny/Ly)^2 + (nz/Lz)^2 ).
Am i right till here? it does make sense!. Also if we had different dimensions what value of n do we have to input? thanks very much for your help so far.
side-note;
I am not a physics student; am a chemistry one. I am sure that QM is same for both but I think you can appreciate that chemists dont touch some of the things that are in the realm of physics.