

sumi
-
Posts
8 -
Joined
-
Last visited
Content Type
Profiles
Forums
Events
Posts posted by sumi
-
-
sue vs use <-- nice oneOops!
Is the point of the exercise to learn to use Chemdraw (or some such) or to learn how to name chemicals?
anyway, tried again with a bit more reading and carefull thinking (only counting hydros that are actually new to basic structures of the naphthalene/bensodioxine rings) and came up with these (I dropped the Stereo, since that's already settled):
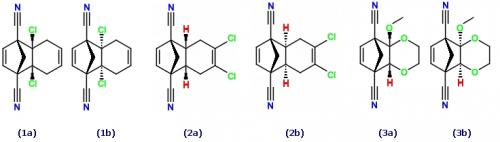
1a+b) 4a,8a-dichloro-5,8-dihydro-1,4-methanonaphthalene-1,4-dicarbonitrile
2a+b) 6,7-dichloro-4a,5,8,8a-tetrahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
3a+b) 4a-methoxy-2,3,8a-trihydro-5,8-methanobenzo[1,4]dioxine-5,8-dicarbonitrile
puh ~ that was a long day(s) work. I'm gonna talk to my prof about these names tomorrow, and see what he thinks of them.
0 -
Not my intention to sue anyone.Is the point of the exercise to learn to sue Chemdraw (or some such) or to learn how to name chemicals?
I was just pointing out why I was using other programs and that they apparently have trouble naming complex molecules correctly
=> thats why I asked for help here ...
0 -
Check your ChemDraw structures are correct. I drew all of your compounds in ChemDraw and got different names to the ones you listed here and the ones you say ChemDraw spat back.
I don't have ChemDraw (does not work on my system), but ChemSketch (ACD)
Very close to the ones you listed above, but your naming of the 'hydro' bits is wrong. Everything else is okay in 1a and b. In 2a and 2b, two of your stereochemistry assignments are wrong. 3a and 3b are not correct either.
Possible Corrections:
1a) (1R,4S,4aS,8aR)-4a,8a-dichloro-1,4,4a,5,8,8a-hexahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
1b) (1R,4S,4aR,8aS)-4a,8a-dichloro-1,4,4a,5,8,8a-hexahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
2a) (1R,4S,4aR,8aS)-6,7-dichloro-1,4,5,6,7,8-hexahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
2b) (1R,4S,4aS,8aR)-6,7-dichloro-1,4,5,6,7,8-hexahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
3a) (4aS,5R,8S,8aR)-4a-methoxy-2,3,4a,5,8,8a-hexahydro-5,8-methanobenzo[1,4]dioxine-5,8-dicarbonitrile
3b) (4aR,5R,8S,8aS)-4a-methoxy-2,3,4a,5,8,8a-hexahydro-5,8-methanobenzo[1,4]dioxine-5,8-dicarbonitrile
0 -
I tried putting the names together myself, now need someone to check them:
1a) (1R,4S,4aS,8aR)-4a,8a-dichloro-2,3,5,5,6,7,8,8-octahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
1b) (1R,4S,4aR,8aS)-4a,8a-dichloro-2,3,5,5,6,7,8,8-octahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
2a) (1R,4S,4aS,8aR)-6,7-dichloro-2,3,4a,5,5,8,8,8a-octahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
2b) (1R,4S,4aR,8aS)-6,7-dichloro-2,3,4a,5,5,8,8,8a-octahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
3a) (1R,4S,4aS,8aR)-4a-methoxy-5,8-dioxa-2,3,6,6,7,7,8a-heptahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
3b) (1R,4S,4aR,8aS)-4a-methoxy-5,8-dioxa-2,3,6,6,7,7,8a-heptahydro-1,4-methanonaphthalene-1,4-dicarbonitrile
Hope someone can help me with this,
sumi
0 -
Are you familiar with the [imath] [x.y.z...n] [/imath] IUPAC naming convention for multicyclics?
Sadly no -.-
Or do you have a copy of the ChemDraw software?I do but both ACD/ChemSketch and ChemAxon/MarvinSketch give me these apparently false names:
(1a) (1R,2S,7R,8S)-4,5-dichlorotricyclo[6.2.1.02,7]undeca-4,9-diene-1,8-dicarbonitrile
(1b) (1R,2R,7S,8S)-4,5-dichlorotricyclo[6.2.1.02,7]undeca-4,9-diene-1,8-dicarbonitrile
(2a) (1R,2S,7R,8S)-2,7-dichlorotricyclo[6.2.1.02,7]undeca-4,9-diene-1,8-dicarbonitrile
(2b) (1R,2R,7S,8S)-2,7-dichlorotricyclo[6.2.1.02,7]undeca-4,9-diene-1,8-dicarbonitrile
(3a) (1R,2S,7R,8S)-2-methoxy-3,6-dioxatricyclo[6.2.1.02,7]undec-9-ene-1,8-dicarbonitrile
(3a) (1R,2R,7S,8S)-2-methoxy-3,6-dioxatricyclo[6.2.1.02,7]undec-9-ene-1,8-dicarbonitrile
0 -
Hi, I have a problem figuring out the name of these compounds (see attachment):
.mol files are attached in a .zip file.
Hope someone can help me with this,
sumi
0 -

Help, name these compounds
in Organic Chemistry
Posted
My prof tells me there a more than one way to name these compounds:
both the derviation from the bridged naphtalene/benzodixine ringsystem as well as from the tricyclic undecadiene ringsystem are possible.
Well, that settels that!