-
Posts
52747 -
Joined
-
Last visited
-
Days Won
258
Content Type
Profiles
Forums
Events
Posts posted by swansont
-
-
Just now, martillo said:
In any hot body there are photons present in the interior of the body. I have already said that this is ignored in Kinetic Theory. It is assumed a body that is transmitting thermal energy ("heat") by conductivity through a unidirectional conductor to another colder body. The hotter body has enough energy stored internally as to be able to provide photons for the heat transmission.
Why? What is the mechanism in your model that gives you these photons?
K-1.kg-1.s * K^3 does not give units of heat or heat flux. The temperature factors don’t even cancel
0 -
!
Moderator Note
Your thread was locked. You don’t get to bring it up again.
1 -
1 hour ago, Sensei said:
Instead of you, we have the Cambridge University dictionary:
https://dictionary.cambridge.org/dictionary/english/self-made
"rich and successful as a result of your own work and not because of family money"
It’s a false dichotomy, because there are other ways of getting money.
If you won the lottery, you’d be rich because you won the lottery, not because of your own work or because of family money.
0 -
10 hours ago, martillo said:
The hotter body continuously provides photons.
How? What mechanism gives you photons?
10 hours ago, martillo said:Yes, something is wrong.
Actually in the model:
H = Q = µ(f,T) = [constant].T3
where [constant] = 2.π.K3/(h2.c3.(e-1) = 1,01660413x102 in SI units: K-1.kg-1.s-5 (precision of 8 digits)
So [constant] is well approximated by 300
Actually is H = Q = V.[constant].T3
Units still don’t work.
0 -
24 minutes ago, martillo said:
The photons incoming from the hotter body at one side of conductor leave it to the colder body at the other side.
So they are only in the material for a short time. How do you get new photons to take their place?
24 minutes ago, martillo said:In the model H = Q = VT3
Heat capacity at constant volume V is defined as:
C = dQ/dT = dH/dT at constant volume V for for dT = 1º KC = 3V(dT)2
Then the capacity per unity of volume
CV = C/V = 3(dT)2 for dT= 1º K
CV = 3 the same as in the Debye model
Your units don’t work. VT^3 doesn’t have the units of Q
0 -
Does your model account for the heat capacity of the material?
What happens to the photons?
0 -
6 hours ago, jv1 said:
The very delicate spring gear assembly is calibrated to push clock hand with certain mass at speed of
6 degrees per second .
When another clock with the same calibration is moving at speed -V1
The momentum of clock hand
increases.
The momentum of gears inside the clock mechanism is increased.
Finally tuned force on clock wound spring is not capable to push clock hand at speed of 6 degrees per sec.
The hand of clock moves slower.Who measures time in such experiments with a spring-driven clock with gears? Mechanical clocks lack the necessary precision.
But time dilation is symmetric. Clock 1 will run slow compared to clock 2, from clock 2”s perspective. How can a mechanical effect have a clock run both fast and slow? It can’t. Time dilation isn’t a mechanical effect
It sounds like you think there is an absolute rest frame.
0 -
I just moved, and am a few hours outside the totality region, but was too occupied to arrange to get inside the zone. And am too wiped out to contemplate travel at this point.
0 -
3 hours ago, martillo said:
When I touch hot water conduction takes place. S-B law does not apply in conduction, it only applies in radiation, I know
Progress
1 -
25 minutes ago, martillo said:
I was wrong in that statement and I admitted it well correcting the thing. I after said that conductivity is actually accomplished in solid metal conductors by the absorption with posterior emission of photons in a process that has a time delay involved and so on and we have a long discussion about. I'm wondering why you aren't you considering all that now. By the way, I know I make mistakes sometimes but always try to correct them.
That’s still radiation, and I’m talking about conduction transfer from one material to another, not through a material. When two materials touch the heat transfer rate is much larger than if they are separated by a small amount (especially if that is a vacuum gap) This is inconsistent with the heat transfer being radiative.
You burn yourself when you touch the hot water. What does the S-B law say is the heat flow from touching your 1 cm^2 fingertip to the coffee?
0 -
On 3/23/2024 at 8:56 AM, martillo said:
The "heat" transferring between two systems is always through the interchange of photons which can pass through the walls separating them. Photons can pass through atoms and molecules (with scattering dispersion sometimes) carrying energy.
This is your claim - heat transfer is from radiation
On 3/15/2024 at 9:58 AM, martillo said:the temperature T (measured by a thermometer) is more related to the energy of the internal EM radiation present in the interior
This is also your claim
0 -
1 minute ago, Sensei said:
Sure. The one who complained about Must and government cooperation..
I don’t know what you mean by cooperation. Government subsidies and tax breaks are not the same as government contracts.
0 -
5 minutes ago, Sensei said:
Isn't it the same as in my example with trading satellites delivered into space for money? i.e. the government pays to lift xxx tons of something for yyy amount of money, instead of NASA.. ?
Sure. Has anyone else brought this up?
0 -
11 minutes ago, Sensei said:
People buy stuff somebody produce..
Without buyers, fortunes cannot grow..
IOW, how can you literally do it yourself if you rely on customers?
Customers trade money for goods and services. They are not just giving money to someone.
12 minutes ago, Sensei said:...if someone has earned several hundred million dollars from PayPal alone
PayPal did not magically appear from nothing.
0 -
1 hour ago, martillo said:
You are right that when we touch the cup conductivity takes place but if not touch no conductivity ad the cup remains radiating heat which can be felt with the face as I mentioned.
But you’ve asserted that there is no conduction, that it’s all radiation. Being inconsistent just makes this worse.
1 hour ago, martillo said:What idea are you referring to? Please explain
YOUR IDEA THAT ALL THERMAL ENERGY AND HEAT TRANSFER IS RADIATION.
1 hour ago, martillo said:Horizontally at some proper distance I can feel the radiation of the coffee cup.
No, you can’t. Not if the cup is cool on the outside.
0 -
1 hour ago, grayson said:
my thought was if an electron absorbed a photon, it could accelerate and have the function of a normal particle accelerator.
Free electrons can’t absorb photons; you can’t conserve both momentum and energy if that were to happen. They scatter photons, giving you a lower-energy photon. (Compton scattering)
You can absorb a photon in the photoelectric effect because the atom is there to let you conserve momentum.
But: acceleration of electrons with light has been done, in various ways
1 -
24 minutes ago, Sensei said:
"Self-made millionaire" means that someone did not get millions from his parents. Heritage them.
"Self-made billionaire" means that someone doesn't get billionaires from his parents.
And that's it.Self-made means you did it yourself, with minimal outside help. Not just help from parents. It suggests that anyone can do it if they just work hard enough.
There’s a baseline of support that anyone can get, but most uber-wealthy get far more than that, such as subsidies or tax breaks for individuals or companies not available to the average person.
If someone hands you a million dollars and you can parlay that into a bigger fortune, good for you. But since most people don’t have access to a million dollars, it’s a tad insulting to imply that it’s a path to wealth accessible to all.
1 -
1 hour ago, martillo said:
Radiation is heat energy transfer rate.
No, that’s the radiation transfer.
You can’t use your idea here, that’s circular reasoning. You need empirical evidence to support it. What we’re doing is some basic testing of the idea.
1 hour ago, martillo said:The rate of transmission is what makes the difference for my fingers "burn" or not. You can have lot of heat energy but transmitted at small rate then it would not affect too much while if it is transferred at a high rate it will affect ("burn").
But that transfer rate drops dramatically once you stop touching the coffee, which is not what would happen with radiation.
1 hour ago, martillo said:I can feel the coffee radiation even with my face at some distance. More the distance less it affects of course.
Not that much difference. The radiation is going up from the surface.
As sethoflagos said, that’s convection. The fluid in this case is the air.
0 -
1 hour ago, martillo said:
The hot coffee can "burn" my fingers while the cup does not. There is a not negligible (not so small as you consider) difference in the intensity of the radiation of both.
To quantity the case properly I would need to work with a thermometer here at home. I don't want to do that.Heat, yes. Radiation, no.
You don’t burn yourself by being close to the hot coffee. You have to touch it. That’s not consistent with radiation.
0 -
59 minutes ago, martillo said:
You must review this. We have a very hot radiation in one side (the internal surface off the cup) and a not so hot in the other side (the external surface of the cup) so, the difference must be or reflected or absorbed. there is no other way.
Of course there’s another way: the radiation level is much lower than you think it is. You don’t quantify anything, so you’re not comparing the numbers that would show this.
The net amount of radiation is actually quite small near room temperature. The heat transfer is via conduction, which is relatively slow, because it depends on the vibration of the atoms.
0 -
34 minutes ago, martillo said:
When we fill the cup it takes some time to its external surface to be heated what is what we can perceive with our fingers. The cup is being heated from its internal surface and it begins to absorb some small percentage of the radiation. While absorbing the heat radiation the heat does not reach the outer surface.
If it only absorbs a small percentage, the rest must be transmitted. We already know the transmission is quite high, so this radiation is not being absorbed.
But we also know the heat in not being transmitted nearly as quickly as this prediction
0 -
7 hours ago, martillo said:
I'm losing the real subject of all of our discussion. Can you please summarize it?
If your hypothesis is true it must be true for all solids. I only have to find one example where it fails.
A material that transmits radiation in the thermal range does so almost instantaneously. (for 2 cm, this was about 0.1 nanoseconds) It must do so regardless of whether the radiation comes from a thermal or non-thermal source, because photons are photons.
But you agree that heat is transmitted much more slowly through such materials. I contend that this falsifies your model. Zinc Selenide does not behave the way you predict it should.
I await your next tap-dance
6 hours ago, martillo said:My model can be compatible with that
You don’t quantify things, so you don’t have a model. If you did the math (or comprehend that math I’ve done for you) you would see that your conjecture is not compatible.
0 -
47 minutes ago, martillo said:
It shows how glass stopped transmitting sharply at about the 3000 nm (3 microns)
That particular glass. The ones I cited transmit out past 10 microns.
http://rmico.com/znse-specifications
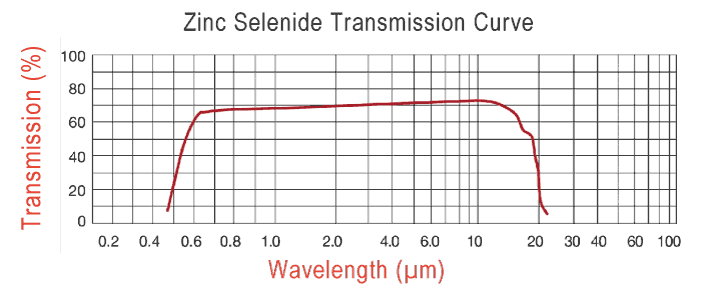 0
0 -
3 minutes ago, Lucas Bet said:
I am fully aware this is a philosophical approach to physics, but requiring evidence? We are in the "Speculations" forum!
You should check the rules of the speculations section.
The first one is
- Speculations must be backed up by evidence or some sort of proof. If your speculation is untestable, or you don't give us evidence (or a prediction that is testable), your thread will be moved to the Trash Can. If you expect any scientific input, you need to provide a case that science can measure.
0



Hypothesis about temperature (split from Physical mechanism how matter absorbs radiation.)
in Speculations
Posted
That’s not a mechanism for generating photons.
No. The Kelvin is a fundamental unit. It does not equate to joules.
Your link does not say what you claim here; you have omitted a factor (in the denominator) of the Boltzmann constant, which has units.